Elfar Adalsteinsson
Massachusetts Institute of Technology, Harvard-MIT Health Sciences and Technology, Institute for Medical Engineering and Science
Scan Specific Artifact Reduction in K-space Neural Networks Synergize with Physics-based Reconstruction to Accelerate MRI
Apr 02, 2021
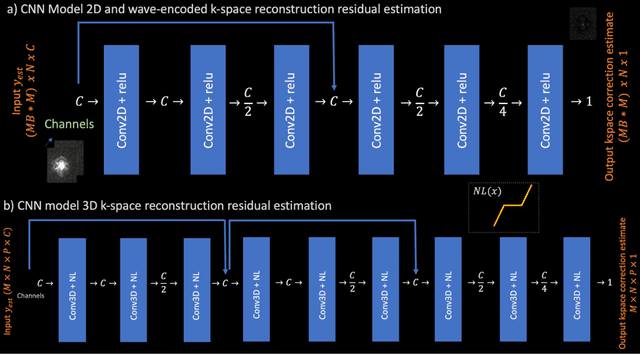
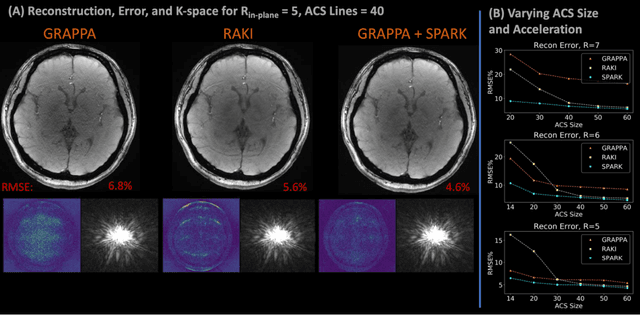
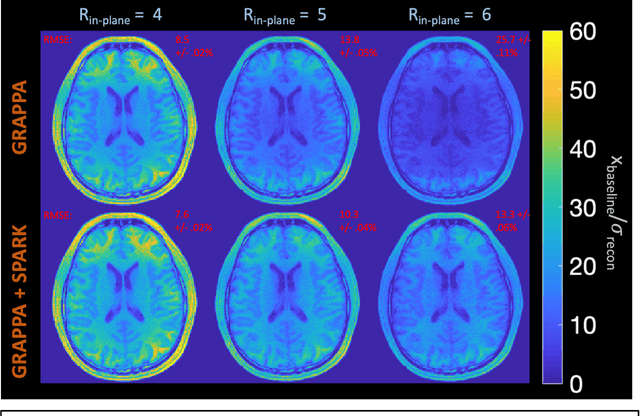
Abstract:Purpose: To develop a scan-specific model that estimates and corrects k-space errors made when reconstructing accelerated Magnetic Resonance Imaging (MRI) data. Methods: Scan-Specific Artifact Reduction in k-space (SPARK) trains a convolutional neural network to estimate k-space errors made by an input reconstruction technique by back-propagating from the mean-squared-error loss between an auto-calibration signal (ACS) and the input technique's reconstructed ACS. First, SPARK is applied to GRAPPA and demonstrates improved robustness over other scan-specific models. Then, SPARK is shown to synergize with advanced reconstruction techniques by improving image quality when applied to 2D virtual coil (VC-) GRAPPA, 2D LORAKS, 3D GRAPPA without an integrated ACS region, and 2D/3D wave-encoded imaging. Results: SPARK yields 1.5 - 2x RMSE reduction when applied to GRAPPA and improves robustness to ACS size for various acceleration rates in comparison to other scan-specific techniques. When applied to advanced parallel imaging techniques such as 2D VC-GRAPPA and LORAKS, SPARK achieves up to 20% RMSE improvement. SPARK with 3D GRAPPA also improves RMSE performance and perceived image quality without a fully sampled ACS region. Finally, SPARK synergizes with non-cartesian, 2D and 3D wave-encoding imaging by reducing RMSE between 20 - 25% and providing qualitative improvements. Conclusion: SPARK synergizes with physics-based reconstruction techniques to improve accelerated MRI by training scan-specific models to estimate and correct reconstruction errors in k-space.
Enhanced detection of fetal pose in 3D MRI by Deep Reinforcement Learning with physical structure priors on anatomy
Jul 16, 2020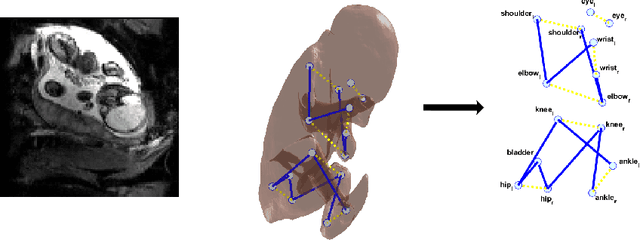
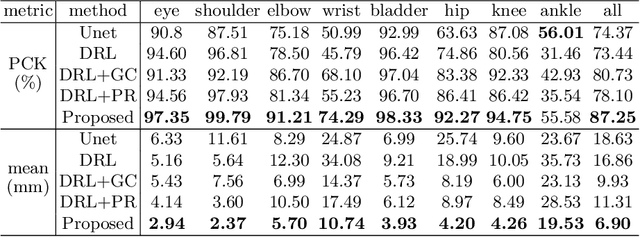
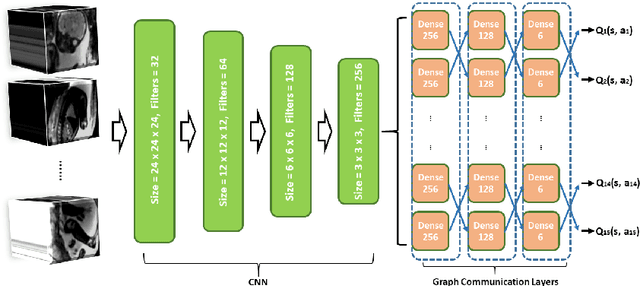
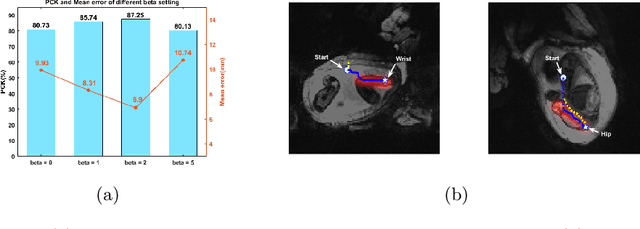
Abstract:Fetal MRI is heavily constrained by unpredictable and substantial fetal motion that causes image artifacts and limits the set of viable diagnostic image contrasts. Current mitigation of motion artifacts is predominantly performed by fast, single-shot MRI and retrospective motion correction. Estimation of fetal pose in real time during MRI stands to benefit prospective methods to detect and mitigate fetal motion artifacts where inferred fetal motion is combined with online slice prescription with low-latency decision making. Current developments of deep reinforcement learning (DRL), offer a novel approach for fetal landmarks detection. In this task 15 agents are deployed to detect 15 landmarks simultaneously by DRL. The optimization is challenging, and here we propose an improved DRL that incorporates priors on physical structure of the fetal body. First, we use graph communication layers to improve the communication among agents based on a graph where each node represents a fetal-body landmark. Further, additional reward based on the distance between agents and physical structures such as the fetal limbs is used to fully exploit physical structure. Evaluation of this method on a repository of 3-mm resolution in vivo data demonstrates a mean accuracy of landmark estimation within 10 mm of ground truth as 87.3%, and a mean error of 6.9 mm. The proposed DRL for fetal pose landmark search demonstrates a potential clinical utility for online detection of fetal motion that guides real-time mitigation of motion artifacts as well as health diagnosis during MRI of the pregnant mother.
Joint Frequency- and Image-Space Learning for Fourier Imaging
Jul 02, 2020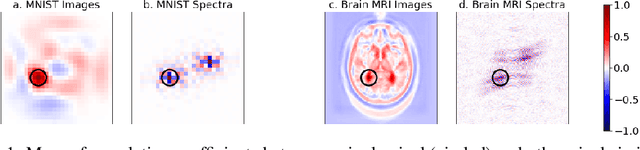
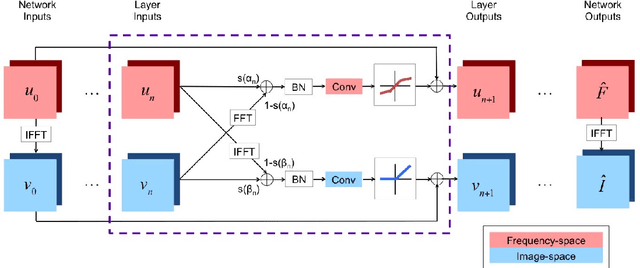
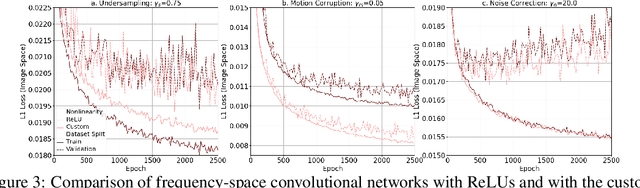
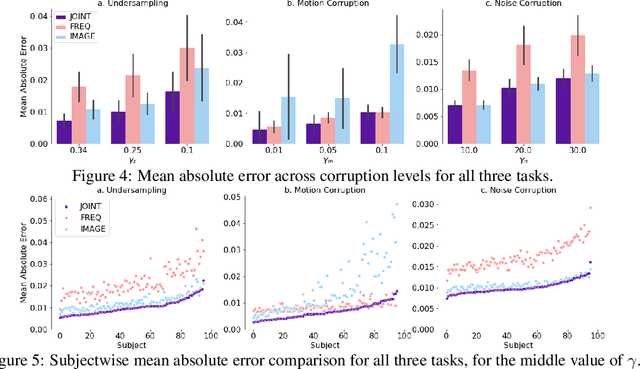
Abstract:We propose a neural network layer structure that combines frequency and image feature representations for robust Fourier image reconstruction. Our work is motivated by the challenges in magnetic resonance imaging (MRI) where the acquired signal is a corrupted Fourier transform of the desired image. The proposed layer structure enables both correction of artifacts native to the frequency-space and manipulation of image-space representations to reconstruct coherent image structures. This is in contrast to the current deep learning approaches for image reconstruction that manipulate data solely in the frequency-space or solely in the image-space. We demonstrate the advantages of the proposed joint learning on three diverse tasks including image reconstruction from undersampled acquisitions, motion correction, and image denoising in brain MRI. Unlike purely image based and purely frequency based architectures, the proposed joint model produces consistently high quality output images. The resulting joint frequency- and image-space feature representations promise to significantly improve modeling and reconstruction of images acquired in the frequency-space. Our code is available at https://github.com/nalinimsingh/interlacer.
Semi-Supervised Learning for Fetal Brain MRI Quality Assessment with ROI consistency
Jun 23, 2020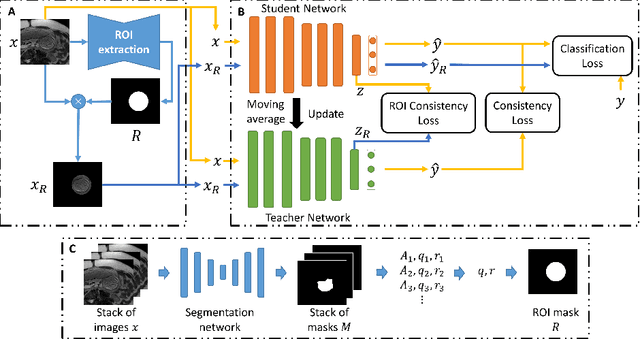
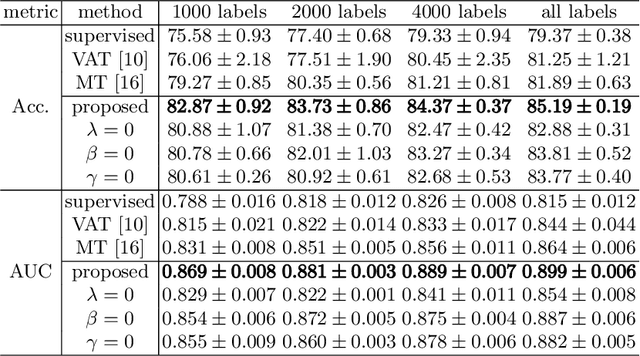
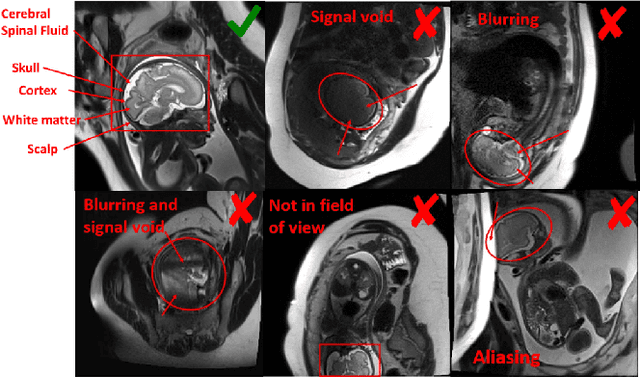
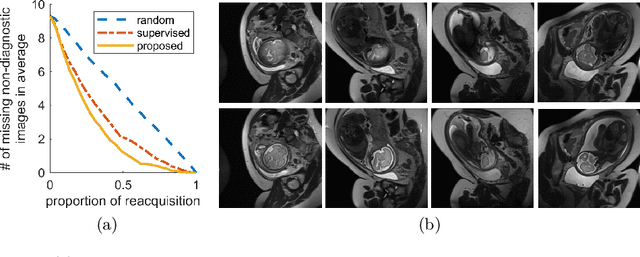
Abstract:Fetal brain MRI is useful for diagnosing brain abnormalities but is challenged by fetal motion. The current protocol for T2-weighted fetal brain MRI is not robust to motion so image volumes are degraded by inter- and intra- slice motion artifacts. Besides, manual annotation for fetal MR image quality assessment are usually time-consuming. Therefore, in this work, a semi-supervised deep learning method that detects slices with artifacts during the brain volume scan is proposed. Our method is based on the mean teacher model, where we not only enforce consistency between student and teacher models on the whole image, but also adopt an ROI consistency loss to guide the network to focus on the brain region. The proposed method is evaluated on a fetal brain MR dataset with 11,223 labeled images and more than 200,000 unlabeled images. Results show that compared with supervised learning, the proposed method can improve model accuracy by about 6\% and outperform other state-of-the-art semi-supervised learning methods. The proposed method is also implemented and evaluated on an MR scanner, which demonstrates the feasibility of online image quality assessment and image reacquisition during fetal MR scans.
Nonlinear Dipole Inversion (NDI) enables Quantitative Susceptibility Mapping (QSM) without parameter tuning
Sep 30, 2019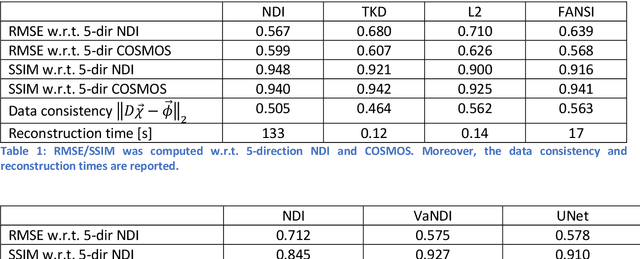
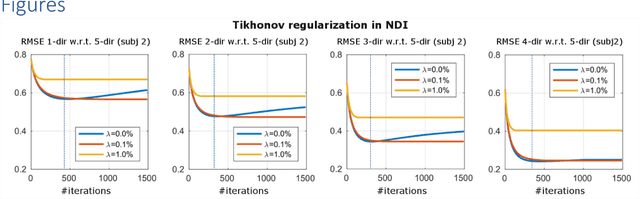
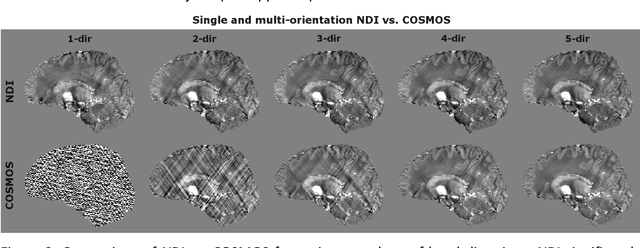
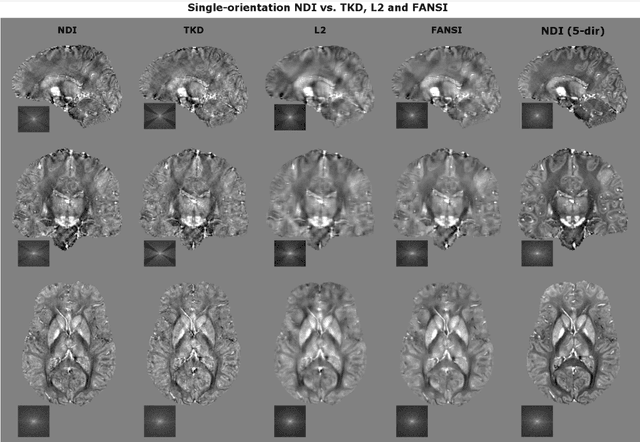
Abstract:We propose Nonlinear Dipole Inversion (NDI) for high-quality Quantitative Susceptibility Mapping (QSM) without regularization tuning, while matching the image quality of state-of-the-art reconstruction techniques. In addition to avoiding over-smoothing that these techniques often suffer from, we also obviate the need for parameter selection. NDI is flexible enough to allow for reconstruction from an arbitrary number of head orientations, and outperforms COSMOS even when using as few as 1-direction data. This is made possible by a nonlinear forward-model that uses the magnitude as an effective prior, for which we derived a simple gradient descent update rule. We synergistically combine this physics-model with a Variational Network (VN) to leverage the power of deep learning in the VaNDI algorithm. This technique adopts the simple gradient descent rule from NDI and learns the network parameters during training, hence requires no additional parameter tuning. Further, we evaluate NDI at 7T using highly accelerated Wave-CAIPI acquisitions at 0.5 mm isotropic resolution and demonstrate high-quality QSM from as few as 2-direction data.
Fetal Pose Estimation in Volumetric MRI using a 3D Convolution Neural Network
Jul 10, 2019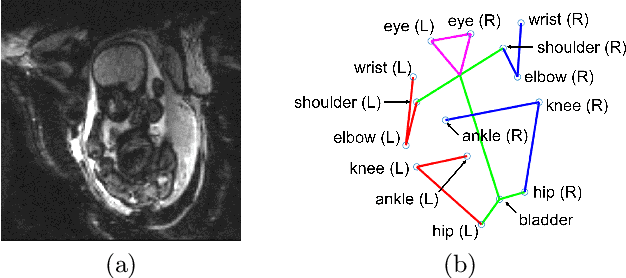
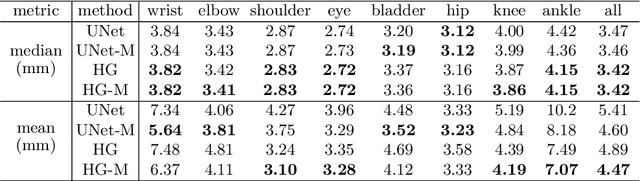
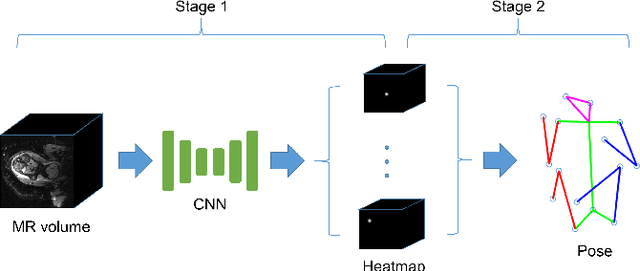
Abstract:The performance and diagnostic utility of magnetic resonance imaging (MRI) in pregnancy is fundamentally constrained by fetal motion. Motion of the fetus, which is unpredictable and rapid on the scale of conventional imaging times, limits the set of viable acquisition techniques to single-shot imaging with severe compromises in signal-to-noise ratio and diagnostic contrast, and frequently results in unacceptable image quality. Surprisingly little is known about the characteristics of fetal motion during MRI and here we propose and demonstrate methods that exploit a growing repository of MRI observations of the gravid abdomen that are acquired at low spatial resolution but relatively high temporal resolution and over long durations (10-30 minutes). We estimate fetal pose per frame in MRI volumes of the pregnant abdomen via deep learning algorithms that detect key fetal landmarks. Evaluation of the proposed method shows that our framework achieves quantitatively an average error of 4.47 mm and 96.4\% accuracy (with error less than 10 mm). Fetal pose estimation in MRI time series yields novel means of quantifying fetal movements in health and disease, and enables the learning of kinematic models that may enhance prospective mitigation of fetal motion artifacts during MRI acquisition.
Temporal Registration in Application to In-utero MRI Time Series
Mar 06, 2019
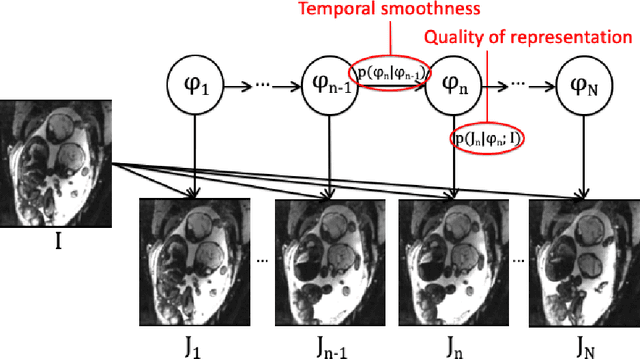
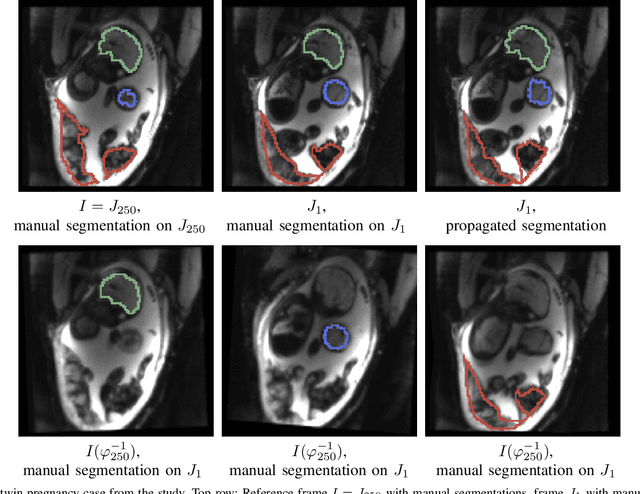
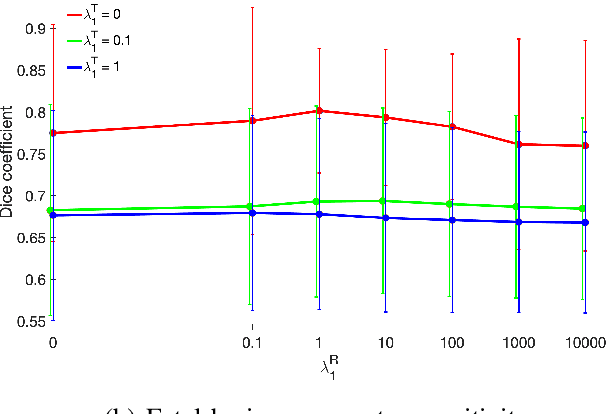
Abstract:We present a robust method to correct for motion in volumetric in-utero MRI time series. Time-course analysis for in-utero volumetric MRI time series often suffers from substantial and unpredictable fetal motion. Registration provides voxel correspondences between images and is commonly employed for motion correction. Current registration methods often fail when aligning images that are substantially different from a template (reference image). To achieve accurate and robust alignment, we make a Markov assumption on the nature of motion and take advantage of the temporal smoothness in the image data. Forward message passing in the corresponding hidden Markov model (HMM) yields an estimation algorithm that only has to account for relatively small motion between consecutive frames. We evaluate the utility of the temporal model in the context of in-utero MRI time series alignment by examining the accuracy of propagated segmentation label maps. Our results suggest that the proposed model captures accurately the temporal dynamics of transformations in in-utero MRI time series.
Temporal Registration in In-Utero Volumetric MRI Time Series
Aug 12, 2016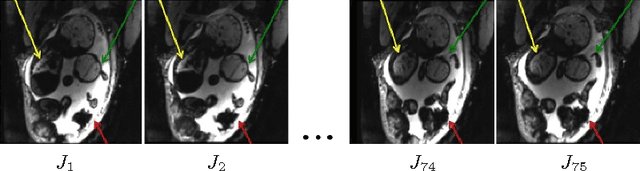
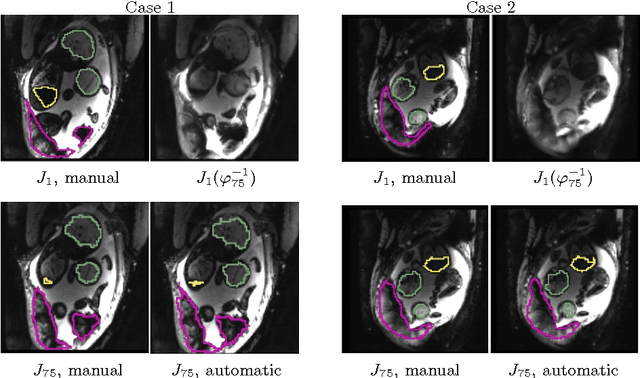
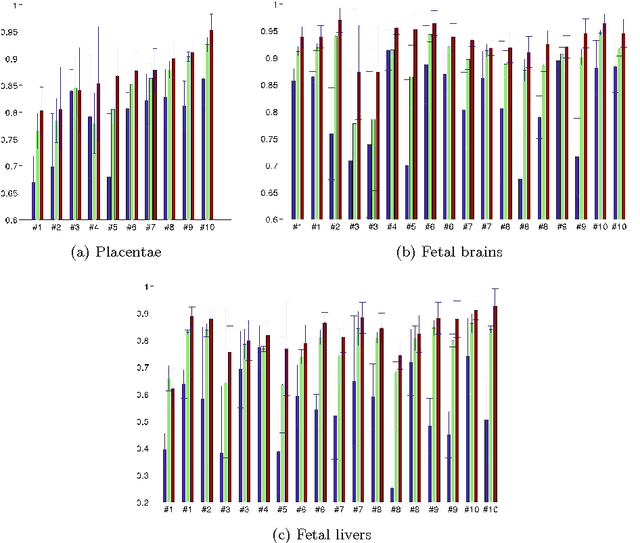
Abstract:We present a robust method to correct for motion and deformations for in-utero volumetric MRI time series. Spatio-temporal analysis of dynamic MRI requires robust alignment across time in the presence of substantial and unpredictable motion. We make a Markov assumption on the nature of deformations to take advantage of the temporal structure in the image data. Forward message passing in the corresponding hidden Markov model (HMM) yields an estimation algorithm that only has to account for relatively small motion between consecutive frames. We demonstrate the utility of the temporal model by showing that its use improves the accuracy of the segmentation propagation through temporal registration. Our results suggest that the proposed model captures accurately the temporal dynamics of deformations in in-utero MRI time series.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge