Alik S. Widge
Bayesian Inference of Psychometric Variables From Brain and Behavior in Implicit Association Tests
Mar 17, 2026Abstract:Objective. We establish a principled method for inferring mental health related psychometric variables from neural and behavioral data using the Implicit Association Test (IAT) as the data generation engine, aiming to overcome the limited predictive performance (typically under 0.7 AUC) of the gold-standard D-score method, which relies solely on reaction times. Approach. We propose a sparse hierarchical Bayesian model that leverages multi-modal data to predict experiences related to mental illness symptoms in new participants. The model is a multivariate generalization of the D-score with trainable parameters, engineered for parameter efficiency in the small-cohort regime typical of IAT studies. Data from two IAT variants were analyzed: a suicidality-related E-IAT ($n=39$) and a psychosis-related PSY-IAT ($n=34$). Main Results. Our approach overcomes a high inter-individual variability and low within-session effect size in the dataset, reaching AUCs of 0.73 (E-IAT) and 0.76 (PSY-IAT) in the best modality configurations, though corrected 95% confidence intervals are wide ($\pm 0.18$) and results are marginally significant after FDR correction ($q=0.10$). Restricting the E-IAT to MDD participants improves AUC to 0.79 $[0.62, 0.97]$ (significant at $q=0.05$). Performance is on par with the best reference methods (shrinkage LDA and EEGNet) for each task, even when the latter were adapted to the task, while the proposed method was not. Accuracy was substantially above near-chance D-scores (0.50-0.53 AUC) in both tasks, with more consistent cross-task performance than any single reference method. Significance. Our framework shows promise for enhancing IAT-based assessment of experiences related to entrapment and psychosis, and potentially other mental health conditions, though further validation on larger and independent cohorts will be needed to establish clinical utility.
Latent Variable Double Gaussian Process Model for Decoding Complex Neural Data
May 08, 2024Abstract:Non-parametric models, such as Gaussian Processes (GP), show promising results in the analysis of complex data. Their applications in neuroscience data have recently gained traction. In this research, we introduce a novel neural decoder model built upon GP models. The core idea is that two GPs generate neural data and their associated labels using a set of low-dimensional latent variables. Under this modeling assumption, the latent variables represent the underlying manifold or essential features present in the neural data. When GPs are trained, the latent variable can be inferred from neural data to decode the labels with a high accuracy. We demonstrate an application of this decoder model in a verbal memory experiment dataset and show that the decoder accuracy in predicting stimulus significantly surpasses the state-of-the-art decoder models. The preceding performance of this model highlights the importance of utilizing non-parametric models in the analysis of neuroscience data.
A 16-Channel Low-Power Neural Connectivity Extraction and Phase-Locked Deep Brain Stimulation SoC
Jul 03, 2022
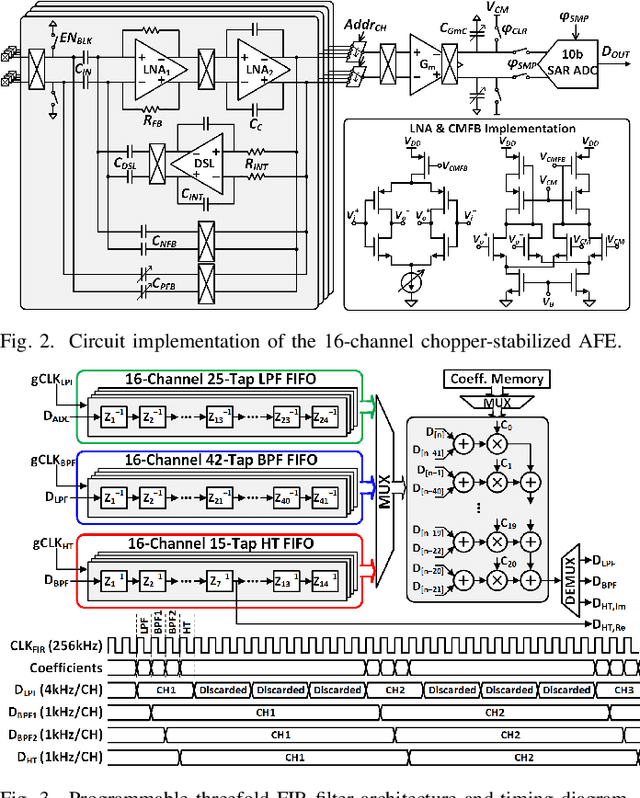
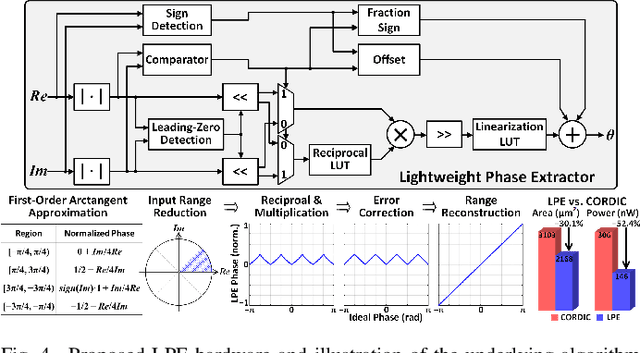
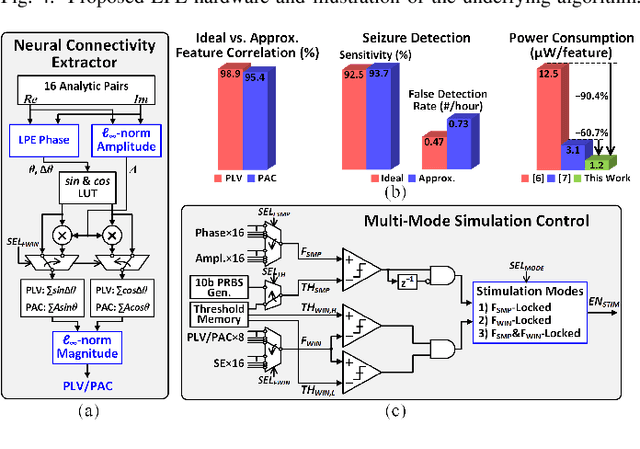
Abstract:Growing evidence suggests that phase-locked deep brain stimulation (DBS) can effectively regulate abnormal brain connectivity in neurological and psychiatric disorders. This letter therefore presents a low-power SoC with both neural connectivity extraction and phase-locked DBS capabilities. A 16-channel low-noise analog front-end (AFE) records local field potentials (LFPs) from multiple brain regions with precise gain matching. A novel low-complexity phase estimator and neural connectivity processor subsequently enable energy-efficient, yet accurate measurement of the instantaneous phase and cross-regional synchrony measures. Through flexible combination of neural biomarkers such as phase synchrony and spectral energy, a four-channel charge-balanced neurostimulator is triggered to treat various pathological brain conditions. Fabricated in 65nm CMOS, the SoC occupies a silicon area of 2.24mm2 and consumes 60uW, achieving over 60% power saving in neural connectivity extraction compared to the state-of-the-art. Extensive in-vivo measurements demonstrate multi-channel LFP recording, real-time extraction of phase and neural connectivity measures, and phase-locked stimulation in rats.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge