Adrian Bowling
A Vision-Language Foundation Model for Zero-shot Clinical Collaboration and Automated Concept Discovery in Dermatology
Feb 11, 2026Abstract:Medical foundation models have shown promise in controlled benchmarks, yet widespread deployment remains hindered by reliance on task-specific fine-tuning. Here, we introduce DermFM-Zero, a dermatology vision-language foundation model trained via masked latent modelling and contrastive learning on over 4 million multimodal data points. We evaluated DermFM-Zero across 20 benchmarks spanning zero-shot diagnosis and multimodal retrieval, achieving state-of-the-art performance without task-specific adaptation. We further evaluated its zero-shot capabilities in three multinational reader studies involving over 1,100 clinicians. In primary care settings, AI assistance enabled general practitioners to nearly double their differential diagnostic accuracy across 98 skin conditions. In specialist settings, the model significantly outperformed board-certified dermatologists in multimodal skin cancer assessment. In collaborative workflows, AI assistance enabled non-experts to surpass unassisted experts while improving management appropriateness. Finally, we show that DermFM-Zero's latent representations are interpretable: sparse autoencoders unsupervisedly disentangle clinically meaningful concepts that outperform predefined-vocabulary approaches and enable targeted suppression of artifact-induced biases, enhancing robustness without retraining. These findings demonstrate that a foundation model can provide effective, safe, and transparent zero-shot clinical decision support.
Revamping AI Models in Dermatology: Overcoming Critical Challenges for Enhanced Skin Lesion Diagnosis
Nov 02, 2023



Abstract:The surge in developing deep learning models for diagnosing skin lesions through image analysis is notable, yet their clinical black faces challenges. Current dermatology AI models have limitations: limited number of possible diagnostic outputs, lack of real-world testing on uncommon skin lesions, inability to detect out-of-distribution images, and over-reliance on dermoscopic images. To address these, we present an All-In-One \textbf{H}ierarchical-\textbf{O}ut of Distribution-\textbf{C}linical Triage (HOT) model. For a clinical image, our model generates three outputs: a hierarchical prediction, an alert for out-of-distribution images, and a recommendation for dermoscopy if clinical image alone is insufficient for diagnosis. When the recommendation is pursued, it integrates both clinical and dermoscopic images to deliver final diagnosis. Extensive experiments on a representative cutaneous lesion dataset demonstrate the effectiveness and synergy of each component within our framework. Our versatile model provides valuable decision support for lesion diagnosis and sets a promising precedent for medical AI applications.
Out-of-Distribution Detection for Long-tailed and Fine-grained Skin Lesion Images
Jun 30, 2022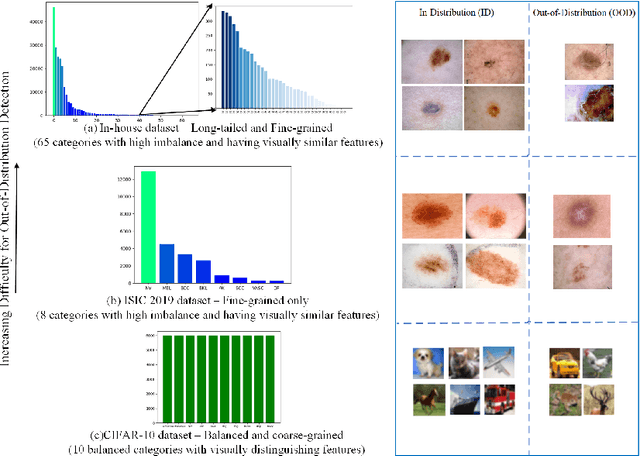
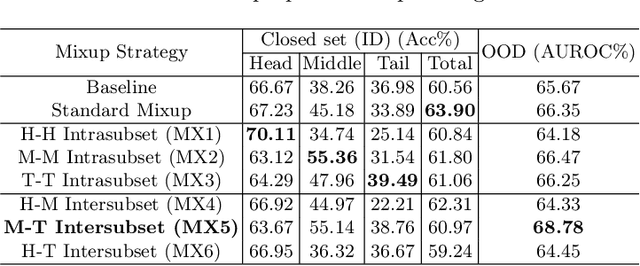
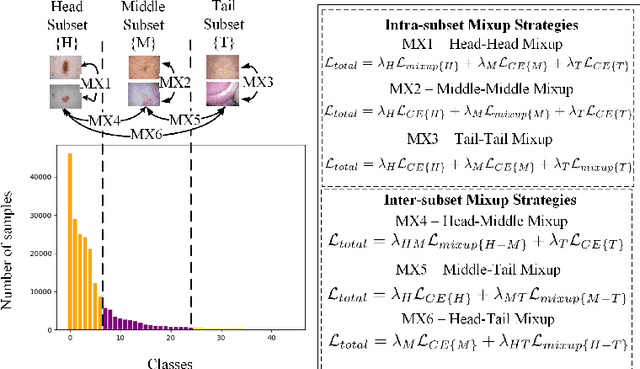
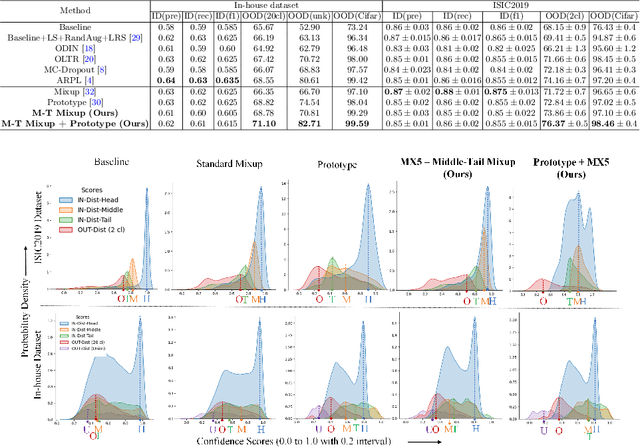
Abstract:Recent years have witnessed a rapid development of automated methods for skin lesion diagnosis and classification. Due to an increasing deployment of such systems in clinics, it has become important to develop a more robust system towards various Out-of-Distribution(OOD) samples (unknown skin lesions and conditions). However, the current deep learning models trained for skin lesion classification tend to classify these OOD samples incorrectly into one of their learned skin lesion categories. To address this issue, we propose a simple yet strategic approach that improves the OOD detection performance while maintaining the multi-class classification accuracy for the known categories of skin lesion. To specify, this approach is built upon a realistic scenario of a long-tailed and fine-grained OOD detection task for skin lesion images. Through this approach, 1) First, we target the mixup amongst middle and tail classes to address the long-tail problem. 2) Later, we combine the above mixup strategy with prototype learning to address the fine-grained nature of the dataset. The unique contribution of this paper is two-fold, justified by extensive experiments. First, we present a realistic problem setting of OOD task for skin lesion. Second, we propose an approach to target the long-tailed and fine-grained aspects of the problem setting simultaneously to increase the OOD performance.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge