Tobias Becher
Tipping the Balance: Impact of Class Imbalance Correction on the Performance of Clinical Risk Prediction Models
Feb 27, 2026Abstract:Objective: ML-based clinical risk prediction models are increasingly used to support decision-making in healthcare. While class-imbalance correction techniques are commonly applied to improve model performance in settings with rare outcomes, their impact on probabilistic calibration remains insufficiently understood. This study evaluated the effect of widely used resampling strategies on both discrimination and calibration across real-world clinical prediction tasks. Methods: Ten clinical datasets spanning diverse medical domains and including 605,842 patients were analyzed. Multiple machine-learning model families, including linear models and several non-linear approaches, were evaluated. Models were trained on the original data and under three commonly used 1:1 class-imbalance correction strategies (SMOTE, RUS, ROS). Performance was assessed on held-out data using discrimination and calibration metrics. Results: Across all datasets and model families, resampling had no positive impact on predictive performance. Changes in the Receiver Operating Characteristic Area Under Curve (ROC-AUC) relative to models trained on the original data were small and inconsistent (ROS: -0.002, p<0.05; RUS: -0.004, p>0.05; SMOTE: -0.01, p<0.05), with no resampling strategy demonstrating a systematic improvement. In contrast, resampling in general degraded the calibration performance. Models trained using imbalance correction exhibited higher Brier scores (0.029 to 0.080, p<0.05), reflecting poorer probabilistic accuracy, and marked deviations in calibration intercept and slope, indicating systematic distortions of predicted risk despite preserved rank-based performance. Conclusion: In a diverse set of real-world clinical prediction tasks, commonly used class-imbalance correction techniques did not provide generalizable improvements in discrimination and were associated with degraded calibration.
Exploring AI-Enabled Cybersecurity Frameworks: Deep-Learning Techniques, GPU Support, and Future Enhancements
Dec 17, 2024



Abstract:Traditional rule-based cybersecurity systems have proven highly effective against known malware threats. However, they face challenges in detecting novel threats. To address this issue, emerging cybersecurity systems are incorporating AI techniques, specifically deep-learning algorithms, to enhance their ability to detect incidents, analyze alerts, and respond to events. While these techniques offer a promising approach to combating dynamic security threats, they often require significant computational resources. Therefore, frameworks that incorporate AI-based cybersecurity mechanisms need to support the use of GPUs to ensure optimal performance. Many cybersecurity framework vendors do not provide sufficiently detailed information about their implementation, making it difficult to assess the techniques employed and their effectiveness. This study aims to overcome this limitation by providing an overview of the most used cybersecurity frameworks that utilize AI techniques, specifically focusing on frameworks that provide comprehensive information about their implementation. Our primary objective is to identify the deep-learning techniques employed by these frameworks and evaluate their support for GPU acceleration. We have identified a total of \emph{two} deep-learning algorithms that are utilized by \emph{three} out of 38 selected cybersecurity frameworks. Our findings aim to assist in selecting open-source cybersecurity frameworks for future research and assessing any discrepancies between deep-learning techniques used in theory and practice.
Inferring respiratory and circulatory parameters from electrical impedance tomography with deep recurrent models
Oct 19, 2020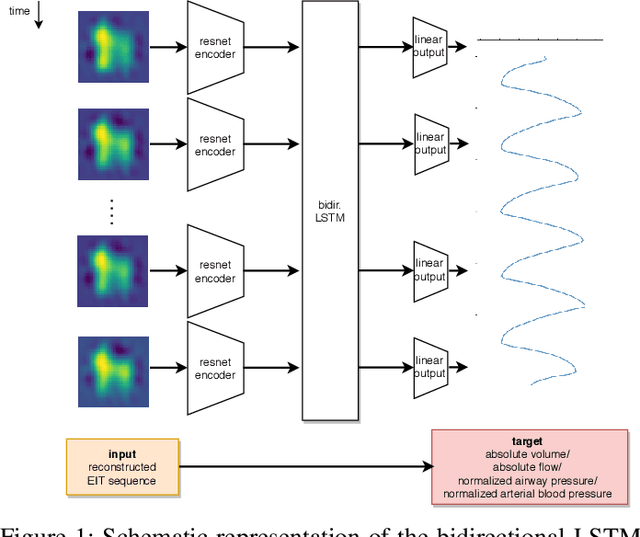
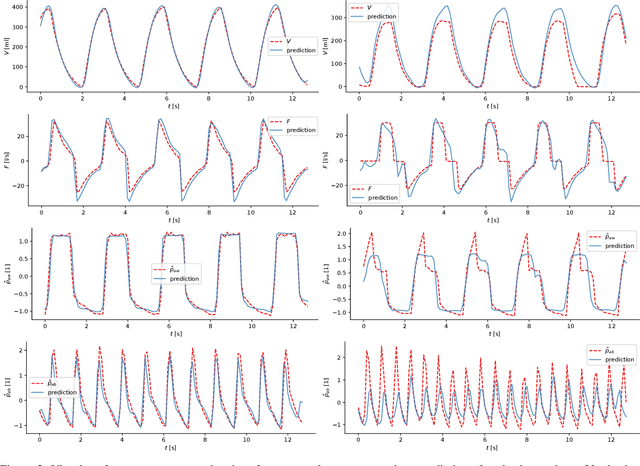
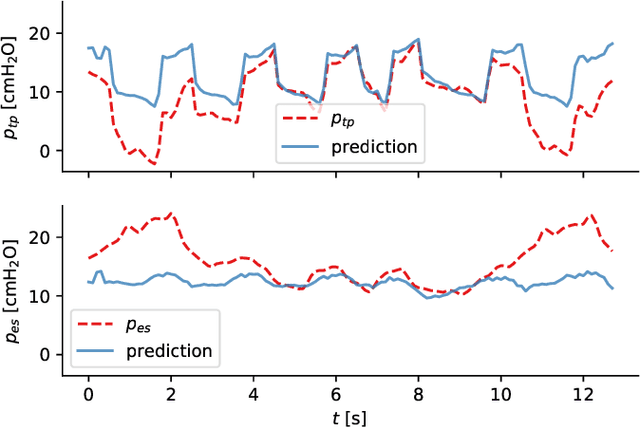
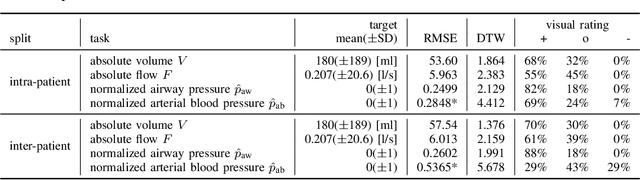
Abstract:Electrical impedance tomography (EIT) is a noninvasive imaging modality that allows a continuous assessment of changes in regional bioimpedance of different organs. One of its most common biomedical applications is monitoring regional ventilation distribution in critically ill patients treated in intensive care units. In this work, we put forward a proof-of-principle study that demonstrates how one can reconstruct synchronously measured respiratory or circulatory parameters from the EIT image sequence using a deep learning model trained in an end-to-end fashion. We demonstrate that one can accurately infer absolute volume, absolute flow, normalized airway pressure and within certain limitations even the normalized arterial blood pressure from the EIT signal alone, in a way that generalizes to unseen patients without prior calibration. As an outlook with direct clinical relevance, we furthermore demonstrate the feasibility of reconstructing the absolute transpulmonary pressure from a combination of EIT and absolute airway pressure, as a way to potentially replace the invasive measurement of esophageal pressure. With these results, we hope to stimulate further studies building on the framework put forward in this work.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge