Ming Jack Po
AnsibleHealth Inc., San Francisco, USA
From Days to Minutes: An Autonomous AI Agent Achieves Reliable Clinical Triage in Remote Patient Monitoring
Mar 10, 2026Abstract:Background: Remote patient monitoring (RPM) generates vast data, yet landmark trials (Tele-HF, BEAT-HF) failed because data volume overwhelmed clinical staff. While TIM-HF2 showed 24/7 physician-led monitoring reduces mortality by 30%, this model remains prohibitively expensive and unscalable. Methods: We developed Sentinel, an autonomous AI agent using Model Context Protocol (MCP) for contextual triage of RPM vitals via 21 clinical tools and multi-step reasoning. Evaluation included: (1) self-consistency (100 readings x 5 runs); (2) comparison against rule-based thresholds; and (3) validation against 6 clinicians (3 physicians, 3 NPs) using a connected matrix design. A leave-one-out (LOO) analysis compared the agent against individual clinicians; severe overtriage cases underwent independent physician adjudication. Results: Against a human majority-vote standard (N=467), the agent achieved 95.8% emergency sensitivity and 88.5% sensitivity for all actionable alerts (85.7% specificity). Four-level exact accuracy was 69.4% (quadratic-weighted kappa=0.778); 95.9% of classifications were within one severity level. In LOO analysis, the agent outperformed every clinician in emergency sensitivity (97.5% vs. 60.0% aggregate) and actionable sensitivity (90.9% vs. 69.5%). While disagreements skewed toward overtriage (22.5%), independent adjudication of severe gaps (>=2 levels) validated agent escalation in 88-94% of cases; consensus resolution validated 100%. The agent showed near-perfect self-consistency (kappa=0.850). Median cost was $0.34/triage. Conclusions: Sentinel triages RPM vitals with sensitivity exceeding individual clinicians. By automating systematic context synthesis, Sentinel addresses the core limitation of prior RPM trials, offering a scalable path toward the intensive monitoring shown to reduce mortality while maintaining a clinically defensible overtriage profile.
Temporal Graph Convolutional Networks for Automatic Seizure Detection
May 03, 2019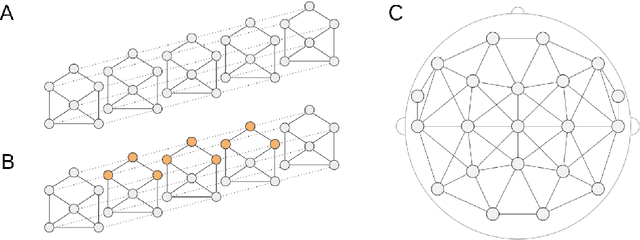
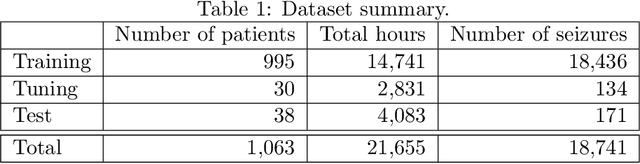
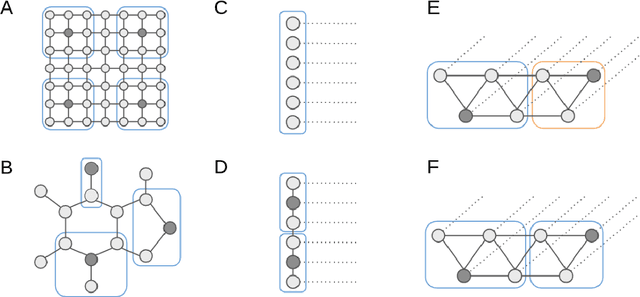
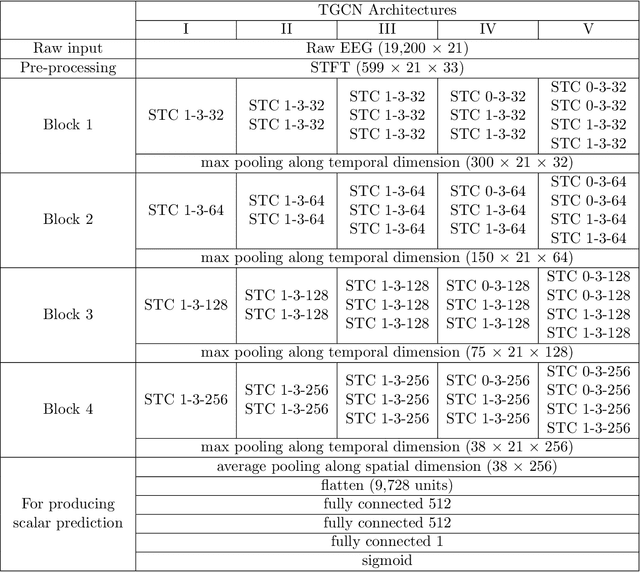
Abstract:Seizure detection from EEGs is a challenging and time consuming clinical problem that would benefit from the development of automated algorithms. EEGs can be viewed as structural time series, because they are multivariate time series where the placement of leads on a patient's scalp provides prior information about the structure of interactions. Commonly used deep learning models for time series don't offer a way to leverage structural information, but this would be desirable in a model for structural time series. To address this challenge, we propose the temporal graph convolutional network (TGCN), a model that leverages structural information and has relatively few parameters. TGCNs apply feature extraction operations that are localized and shared over both time and space, thereby providing a useful inductive bias in tasks where one expects similar features to be discriminative across the different sequences. In our experiments we focus on metrics that are most important to seizure detection, and demonstrate that TGCN matches the performance of related models that have been shown to be state of the art in other tasks. Additionally, we investigate interpretability advantages of TGCN by exploring approaches for helping clinicians determine when precisely seizures occur, and the parts of the brain that are most involved.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge