Juntang Zhuang
Tony
LadderNet: Multi-path networks based on U-Net for medical image segmentation
Oct 27, 2018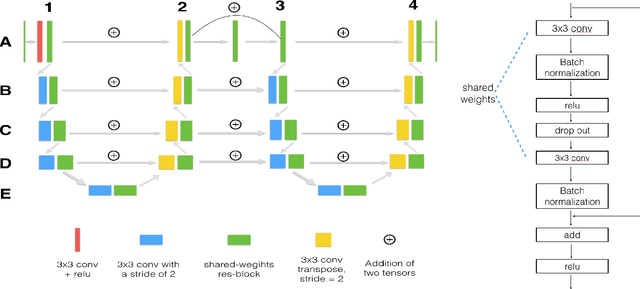
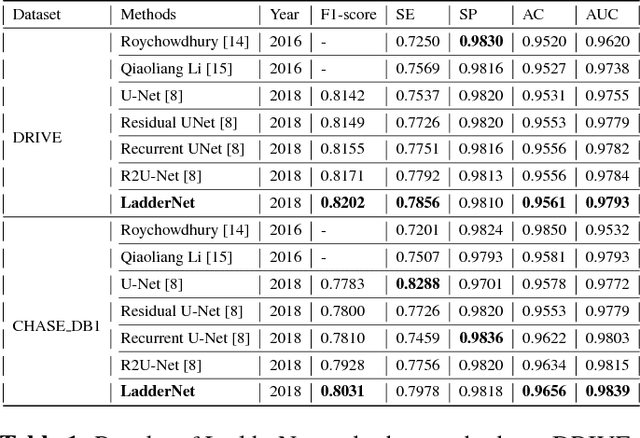
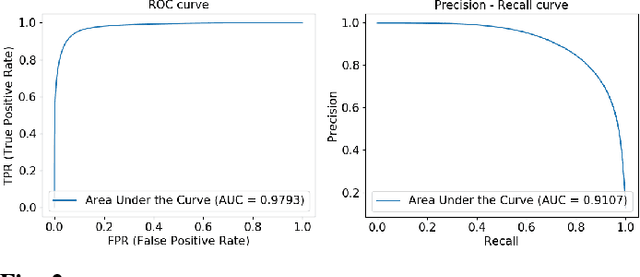
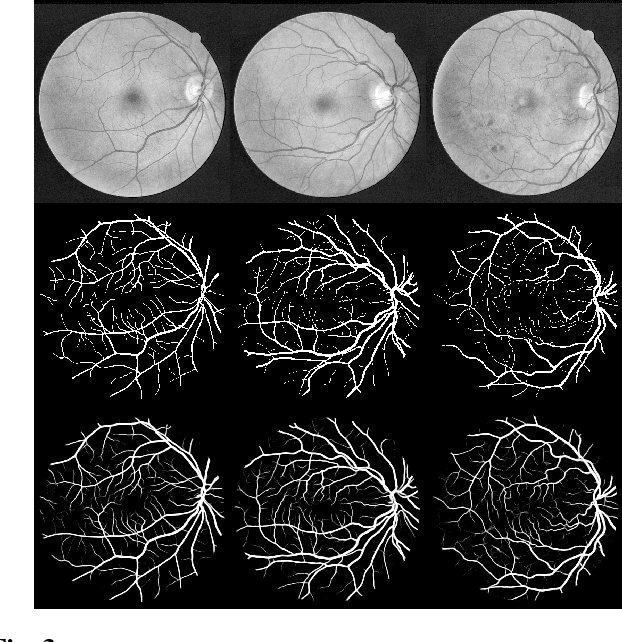
Abstract:U-Net has been providing state-of-the-art performance in many medical image segmentation problems. Many modifications have been proposed for U-Net, such as attention U-Net, recurrent residual convolutional U-Net (R2-UNet), and U-Net with residual blocks or blocks with dense connections. However, all these modifications have an encoder-decoder structure with skip connections, and the number of paths for information flow is limited. We propose LadderNet in this paper, which can be viewed as a chain of multiple U-Nets. Instead of only one pair of encoder branch and decoder branch in U-Net, a LadderNet has multiple pairs of encoder-decoder branches, and has skip connections between every pair of adjacent decoder and decoder branches in each level. Inspired by the success of ResNet and R2-UNet, we use modified residual blocks where two convolutional layers in one block share the same weights. A LadderNet has more paths for information flow because of skip connections and residual blocks, and can be viewed as an ensemble of Fully Convolutional Networks (FCN). The equivalence to an ensemble of FCNs improves segmentation accuracy, while the shared weights within each residual block reduce parameter number. Semantic segmentation is essential for retinal disease detection. We tested LadderNet on two benchmark datasets for blood vessel segmentation in retinal images, and achieved superior performance over methods in the literature.
Brain Biomarker Interpretation in ASD Using Deep Learning and fMRI
Aug 23, 2018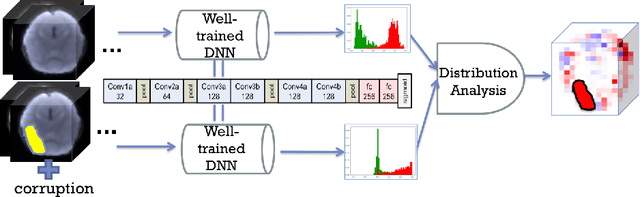

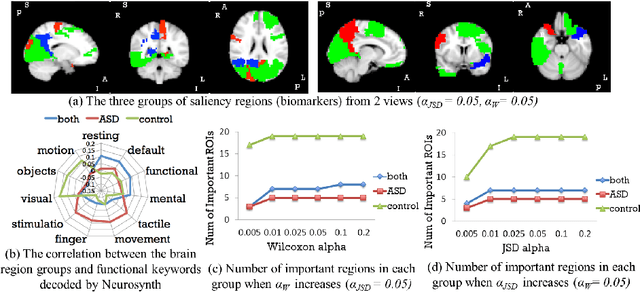
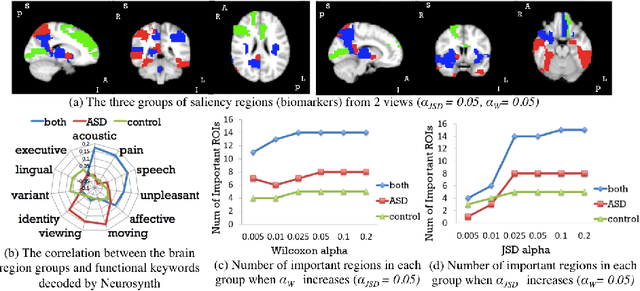
Abstract:Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder. Finding the biomarkers associated with ASD is extremely helpful to understand the underlying roots of the disorder and can lead to earlier diagnosis and more targeted treatment. Although Deep Neural Networks (DNNs) have been applied in functional magnetic resonance imaging (fMRI) to identify ASD, understanding the data-driven computational decision making procedure has not been previously explored. Therefore, in this work, we address the problem of interpreting reliable biomarkers associated with identifying ASD; specifically, we propose a 2-stage method that classifies ASD and control subjects using fMRI images and interprets the saliency features activated by the classifier. First, we trained an accurate DNN classifier. Then, for detecting the biomarkers, different from the DNN visualization works in computer vision, we take advantage of the anatomical structure of brain fMRI and develop a frequency-normalized sampling method to corrupt images. Furthermore, in the ASD vs. control subjects classification scenario, we provide a new approach to detect and characterize important brain features into three categories. The biomarkers we found by the proposed method are robust and consistent with previous findings in the literature. We also validate the detected biomarkers by neurological function decoding and comparing with the DNN activation maps.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge