Giovanni Cinà
Pacmed
Is my model perplexed for the right reason? Contrasting LLMs' Benchmark Behavior with Token-Level Perplexity
Mar 31, 2026Abstract:Standard evaluations of Large language models (LLMs) focus on task performance, offering limited insight into whether correct behavior reflects appropriate underlying mechanisms and risking confirmation bias. We introduce a simple, principled interpretability framework based on token-level perplexity to test whether models rely on linguistically relevant cues. By comparing perplexity distributions over minimal sentence pairs differing in one or a few `pivotal' tokens, our method enables precise, hypothesis-driven analysis without relying on unstable feature-attribution techniques. Experiments on controlled linguistic benchmarks with several open-weight LLMs show that, while linguistically important tokens influence model behavior, they never fully explain perplexity shifts, revealing that models rely on heuristics other than the expected linguistic ones.
A Causal Framework for Evaluating ICU Discharge Strategies
Mar 26, 2026Abstract:In this applied paper, we address the difficult open problem of when to discharge patients from the Intensive Care Unit. This can be conceived as an optimal stopping scenario with three added challenges: 1) the evaluation of a stopping strategy from observational data is itself a complex causal inference problem, 2) the composite objective is to minimize the length of intervention and maximize the outcome, but the two cannot be collapsed to a single dimension, and 3) the recording of variables stops when the intervention is discontinued. Our contributions are two-fold. First, we generalize the implementation of the g-formula Python package, providing a framework to evaluate stopping strategies for problems with the aforementioned structure, including positivity and coverage checks. Second, with a fully open-source pipeline, we apply this approach to MIMIC-IV, a public ICU dataset, demonstrating the potential for strategies that improve upon current care.
Improving TabPFN's Synthetic Data Generation by Integrating Causal Structure
Mar 10, 2026Abstract:Synthetic tabular data generation addresses data scarcity and privacy constraints in a variety of domains. Tabular Prior-Data Fitted Network (TabPFN), a recent foundation model for tabular data, has been shown capable of generating high-quality synthetic tabular data. However, TabPFN is autoregressive: features are generated sequentially by conditioning on the previous ones, depending on the order in which they appear in the input data. We demonstrate that when the feature order conflicts with causal structure, the model produces spurious correlations that impair its ability to generate synthetic data and preserve causal effects. We address this limitation by integrating causal structure into TabPFN's generation process through two complementary approaches: Directed Acyclic Graph (DAG)-aware conditioning, which samples each variable given its causal parents, and a Completed Partially Directed Acyclic Graph (CPDAG)-based strategy for scenarios with partial causal knowledge. We evaluate these approaches on controlled benchmarks and six CSuite datasets, assessing structural fidelity, distributional alignment, privacy preservation, and Average Treatment Effect (ATE) preservation. Across most settings, DAG-aware conditioning improves the quality and stability of synthetic data relative to vanilla TabPFN. The CPDAG-based strategy shows moderate improvements, with effectiveness depending on the number of oriented edges. These results indicate that injecting causal structure into autoregressive generation enhances the reliability of synthetic tabular data.
2-Step Agent: A Framework for the Interaction of a Decision Maker with AI Decision Support
Feb 25, 2026Abstract:Across a growing number of fields, human decision making is supported by predictions from AI models. However, we still lack a deep understanding of the effects of adoption of these technologies. In this paper, we introduce a general computational framework, the 2-Step Agent, which models the effects of AI-assisted decision making. Our framework uses Bayesian methods for causal inference to model 1) how a prediction on a new observation affects the beliefs of a rational Bayesian agent, and 2) how this change in beliefs affects the downstream decision and subsequent outcome. Using this framework, we show by simulations how a single misaligned prior belief can be sufficient for decision support to result in worse downstream outcomes compared to no decision support. Our results reveal several potential pitfalls of AI-driven decision support and highlight the need for thorough model documentation and proper user training.
Mitigating Overconfidence in Out-of-Distribution Detection by Capturing Extreme Activations
May 21, 2024Abstract:Detecting out-of-distribution (OOD) instances is crucial for the reliable deployment of machine learning models in real-world scenarios. OOD inputs are commonly expected to cause a more uncertain prediction in the primary task; however, there are OOD cases for which the model returns a highly confident prediction. This phenomenon, denoted as "overconfidence", presents a challenge to OOD detection. Specifically, theoretical evidence indicates that overconfidence is an intrinsic property of certain neural network architectures, leading to poor OOD detection. In this work, we address this issue by measuring extreme activation values in the penultimate layer of neural networks and then leverage this proxy of overconfidence to improve on several OOD detection baselines. We test our method on a wide array of experiments spanning synthetic data and real-world data, tabular and image datasets, multiple architectures such as ResNet and Transformer, different training loss functions, and include the scenarios examined in previous theoretical work. Compared to the baselines, our method often grants substantial improvements, with double-digit increases in OOD detection AUC, and it does not damage performance in any scenario.
Causal prediction models for medication safety monitoring: The diagnosis of vancomycin-induced acute kidney injury
Nov 15, 2023


Abstract:The current best practice approach for the retrospective diagnosis of adverse drug events (ADEs) in hospitalized patients relies on a full patient chart review and a formal causality assessment by multiple medical experts. This evaluation serves to qualitatively estimate the probability of causation (PC); the probability that a drug was a necessary cause of an adverse event. This practice is manual, resource intensive and prone to human biases, and may thus benefit from data-driven decision support. Here, we pioneer a causal modeling approach using observational data to estimate a lower bound of the PC (PC$_{low}$). This method includes two key causal inference components: (1) the target trial emulation framework and (2) estimation of individualized treatment effects using machine learning. We apply our method to the clinically relevant use-case of vancomycin-induced acute kidney injury in intensive care patients, and compare our causal model-based PC$_{low}$ estimates to qualitative estimates of the PC provided by a medical expert. Important limitations and potential improvements are discussed, and we conclude that future improved causal models could provide essential data-driven support for medication safety monitoring in hospitalized patients.
Fixing confirmation bias in feature attribution methods via semantic match
Jul 03, 2023



Abstract:Feature attribution methods have become a staple method to disentangle the complex behavior of black box models. Despite their success, some scholars have argued that such methods suffer from a serious flaw: they do not allow a reliable interpretation in terms of human concepts. Simply put, visualizing an array of feature contributions is not enough for humans to conclude something about a model's internal representations, and confirmation bias can trick users into false beliefs about model behavior. We argue that a structured approach is required to test whether our hypotheses on the model are confirmed by the feature attributions. This is what we call the "semantic match" between human concepts and (sub-symbolic) explanations. Building on the conceptual framework put forward in Cin\`a et al. [2023], we propose a structured approach to evaluate semantic match in practice. We showcase the procedure in a suite of experiments spanning tabular and image data, and show how the assessment of semantic match can give insight into both desirable (e.g., focusing on an object relevant for prediction) and undesirable model behaviors (e.g., focusing on a spurious correlation). We couple our experimental results with an analysis on the metrics to measure semantic match, and argue that this approach constitutes the first step towards resolving the issue of confirmation bias in XAI.
Semantic match: Debugging feature attribution methods in XAI for healthcare
Jan 06, 2023



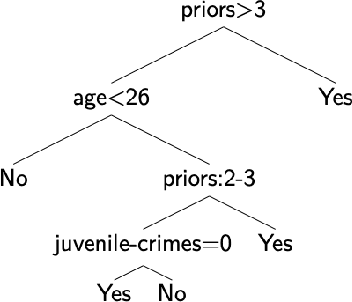
Abstract:The recent spike in certified Artificial Intelligence (AI) tools for healthcare has renewed the debate around adoption of this technology. One thread of such debate concerns Explainable AI (XAI) and its promise to render AI devices more transparent and trustworthy. A few voices active in the medical AI space have expressed concerns on the reliability of Explainable AI techniques and especially feature attribution methods, questioning their use and inclusion in guidelines and standards. Despite valid concerns, we argue that existing criticism on the viability of post-hoc local explainability methods throws away the baby with the bathwater by generalizing a problem that is specific to image data. We begin by characterizing the problem as a lack of semantic match between explanations and human understanding. To understand when feature importance can be used reliably, we introduce a distinction between feature importance of low- and high-level features. We argue that for data types where low-level features come endowed with a clear semantics, such as tabular data like Electronic Health Records (EHRs), semantic match can be obtained, and thus feature attribution methods can still be employed in a meaningful and useful way.
Why we do need Explainable AI for Healthcare
Jun 30, 2022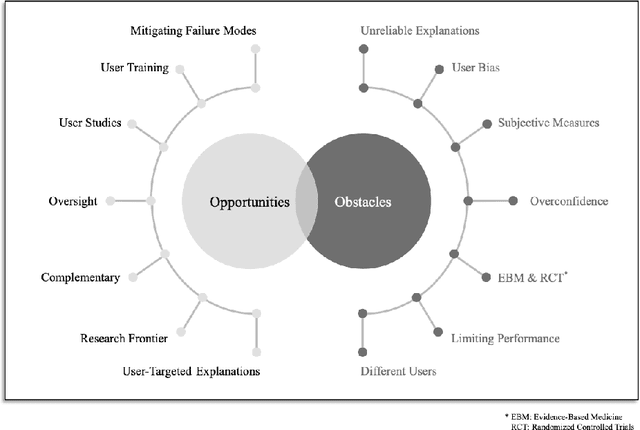
Abstract:The recent spike in certified Artificial Intelligence (AI) tools for healthcare has renewed the debate around adoption of this technology. One thread of such debate concerns Explainable AI and its promise to render AI devices more transparent and trustworthy. A few voices active in the medical AI space have expressed concerns on the reliability of Explainable AI techniques, questioning their use and inclusion in guidelines and standards. Revisiting such criticisms, this article offers a balanced and comprehensive perspective on the utility of Explainable AI, focusing on the specificity of clinical applications of AI and placing them in the context of healthcare interventions. Against its detractors and despite valid concerns, we argue that the Explainable AI research program is still central to human-machine interaction and ultimately our main tool against loss of control, a danger that cannot be prevented by rigorous clinical validation alone.
Out-of-Distribution Detection for Medical Applications: Guidelines for Practical Evaluation
Sep 30, 2021
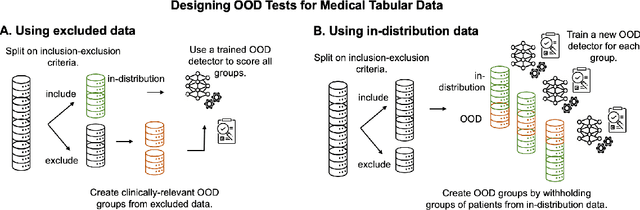
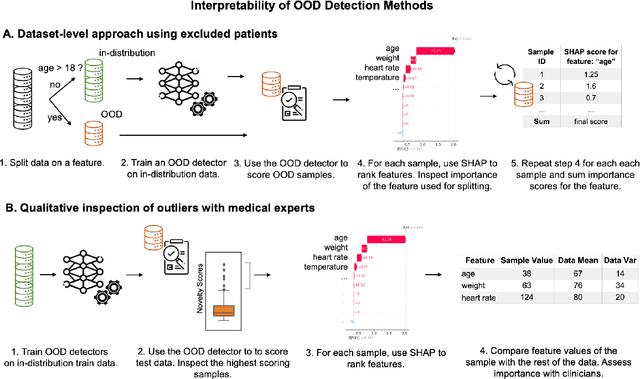
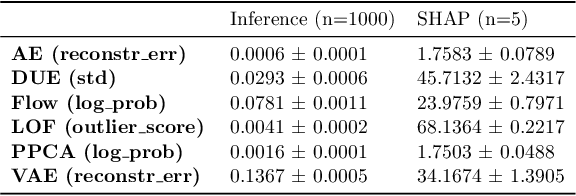
Abstract:Detection of Out-of-Distribution (OOD) samples in real time is a crucial safety check for deployment of machine learning models in the medical field. Despite a growing number of uncertainty quantification techniques, there is a lack of evaluation guidelines on how to select OOD detection methods in practice. This gap impedes implementation of OOD detection methods for real-world applications. Here, we propose a series of practical considerations and tests to choose the best OOD detector for a specific medical dataset. These guidelines are illustrated on a real-life use case of Electronic Health Records (EHR). Our results can serve as a guide for implementation of OOD detection methods in clinical practice, mitigating risks associated with the use of machine learning models in healthcare.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge