Afra Nawar
Guideline2Graph: Profile-Aware Multimodal Parsing for Executable Clinical Decision Graphs
Apr 02, 2026Abstract:Clinical practice guidelines are long, multimodal documents whose branching recommendations are difficult to convert into executable clinical decision support (CDS), and one-shot parsing often breaks cross-page continuity. Recent LLM/VLM extractors are mostly local or text-centric, under-specifying section interfaces and failing to consolidate cross-page control flow across full documents into one coherent decision graph. We present a decomposition-first pipeline that converts full-guideline evidence into an executable clinical decision graph through topology-aware chunking, interface-constrained chunk graph generation, and provenance-preserving global aggregation. Rather than relying on single-pass generation, the pipeline uses explicit entry/terminal interfaces and semantic deduplication to preserve cross-page continuity while keeping the induced control flow auditable and structurally consistent. We evaluate on an adjudicated prostate-guideline benchmark with matched inputs and the same underlying VLM backbone across compared methods. On the complete merged graph, our approach improves edge and triplet precision/recall from $19.6\%/16.1\%$ in existing models to $69.0\%/87.5\%$, while node recall rises from $78.1\%$ to $93.8\%$. These results support decomposition-first, auditable guideline-to-CDS conversion on this benchmark, while current evidence remains limited to one adjudicated prostate guideline and motivates broader multi-guideline validation.
Topological Descriptors for Parkinson's Disease Classification and Regression Analysis
May 06, 2020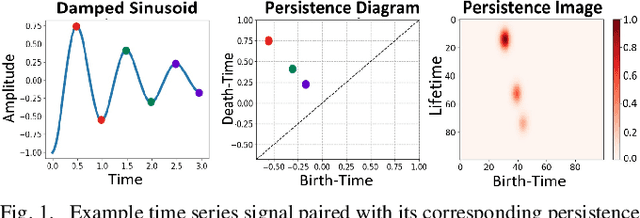
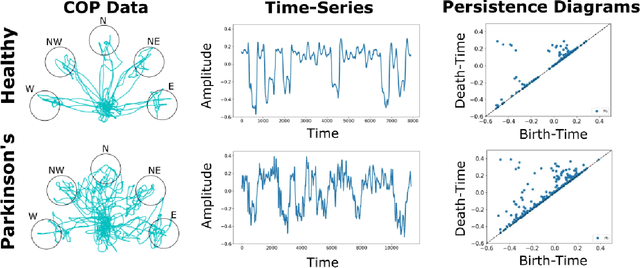
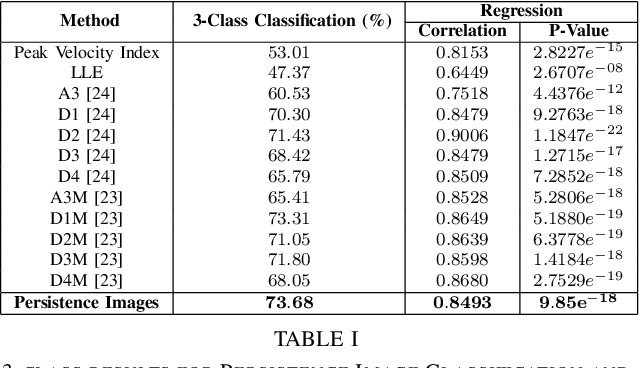
Abstract:At present, the vast majority of human subjects with neurological disease are still diagnosed through in-person assessments and qualitative analysis of patient data. In this paper, we propose to use Topological Data Analysis (TDA) together with machine learning tools to automate the process of Parkinson's disease classification and severity assessment. An automated, stable, and accurate method to evaluate Parkinson's would be significant in streamlining diagnoses of patients and providing families more time for corrective measures. We propose a methodology which incorporates TDA into analyzing Parkinson's disease postural shifts data through the representation of persistence images. Studying the topology of a system has proven to be invariant to small changes in data and has been shown to perform well in discrimination tasks. The contributions of the paper are twofold. We propose a method to 1) classify healthy patients from those afflicted by disease and 2) diagnose the severity of disease. We explore the use of the proposed method in an application involving a Parkinson's disease dataset comprised of healthy-elderly, healthy-young and Parkinson's disease patients. Our code is available at https://github.com/itsmeafra/Sublevel-Set-TDA.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge