Kayvan Najarian
ReDMark: Framework for Residual Diffusion Watermarking on Deep Networks
Oct 16, 2018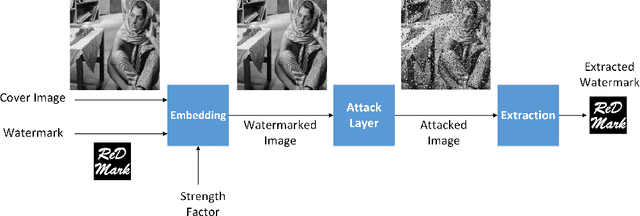
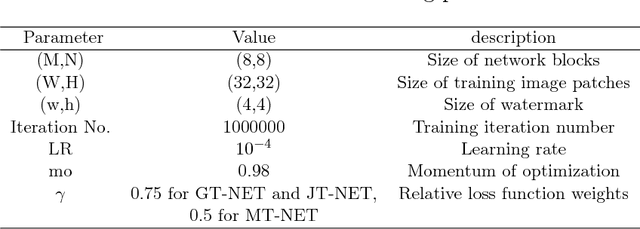
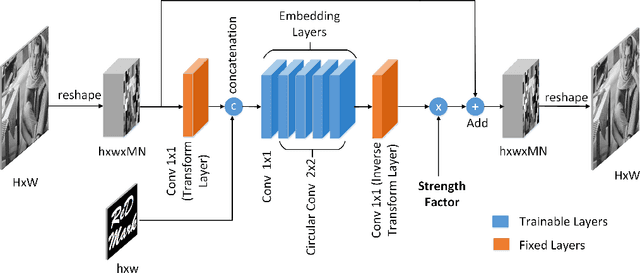
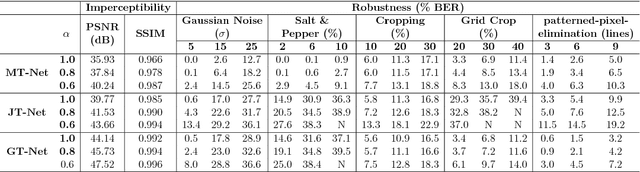
Abstract:Due to the rapid growth of machine learning tools and specifically deep networks in various computer vision and image processing areas, application of Convolutional Neural Networks for watermarking have recently emerged. In this paper, we propose a deep end-to-end diffusion watermarking framework (ReDMark) which can be adapted for any desired transform space. The framework is composed of two Fully Convolutional Neural Networks with the residual structure for embedding and extraction. The whole deep network is trained end-to-end to conduct a blind secure watermarking. The framework is customizable for the level of robustness vs. imperceptibility. It is also adjustable for the trade-off between capacity and robustness. The proposed framework simulates various attacks as a differentiable network layer to facilitate end-to-end training. For JPEG attack, a differentiable approximation is utilized, which drastically improves the watermarking robustness to this attack. Another important characteristic of the proposed framework, which leads to improved security and robustness, is its capability to diffuse watermark information among a relatively wide area of the image. Comparative results versus recent state-of-the-art researches highlight the superiority of the proposed framework in terms of imperceptibility and robustness.
Multiple Abnormality Detection for Automatic Medical Image Diagnosis Using Bifurcated Convolutional Neural Network
Oct 15, 2018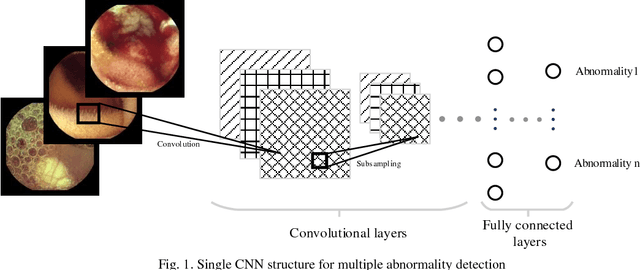
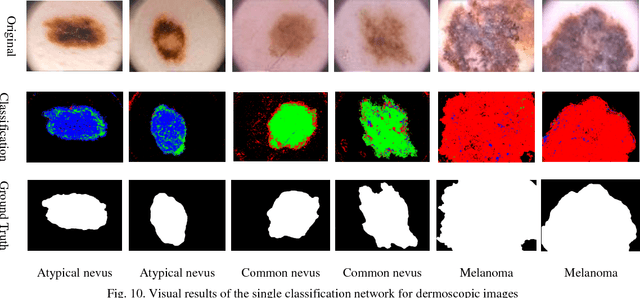
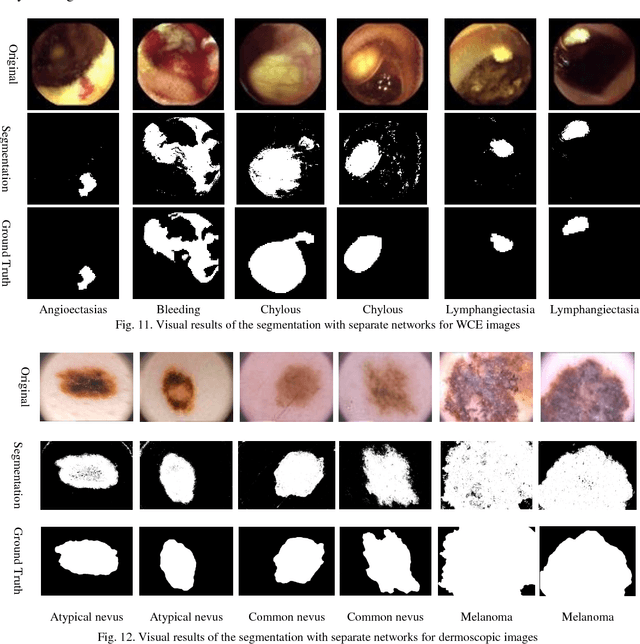
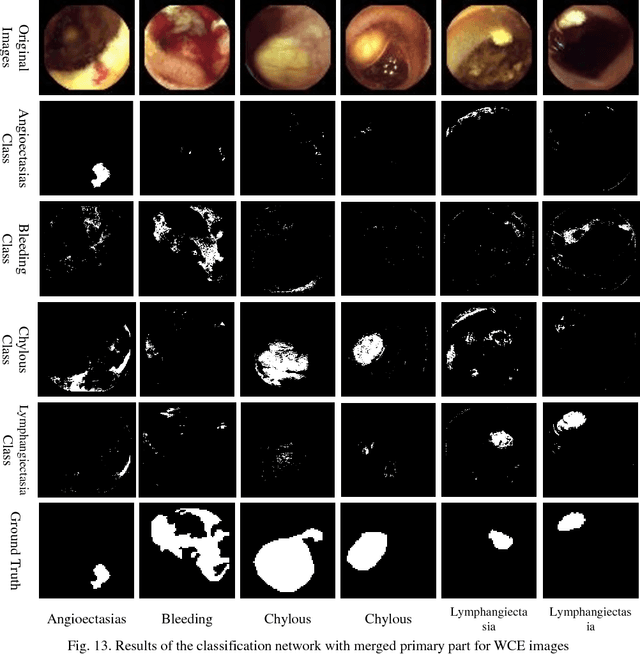
Abstract:Automating classification and segmentation process of abnormal regions in different body organs has a crucial role in most of medical imaging applications such as funduscopy, endoscopy, and dermoscopy. Detecting multiple abnormalities in each type of images is necessary for better and more accurate diagnosis procedure and medical decisions. In recent years portable medical imaging devices such as capsule endoscopy and digital dermatoscope have been introduced and made the diagnosis procedure easier and more efficient. However, these portable devices have constrained power resources and limited computational capability. To address this problem, we propose a bifurcated structure for convolutional neural networks performing both classification and segmentation of multiple abnormalities simultaneously. The proposed network is first trained by each abnormality separately. Then the network is trained using all abnormalities. In order to reduce the computational complexity, the network is redesigned to share some features which are common among all abnormalities. Later, these shared features are used in different settings (directions) to segment and classify the abnormal region of the image. Finally, results of the classification and segmentation directions are fused to obtain the classified segmentation map. Proposed framework is simulated using four frequent gastrointestinal abnormalities as well as three dermoscopic lesions and for evaluation of the proposed framework the results are compared with the corresponding ground truth map. Properties of the bifurcated network like low complexity and resource sharing make it suitable to be implemented as a part of portable medical imaging devices.
Segmentation of Bleeding Regions in Wireless Capsule Endoscopy for Detection of Informative Frames
Aug 23, 2018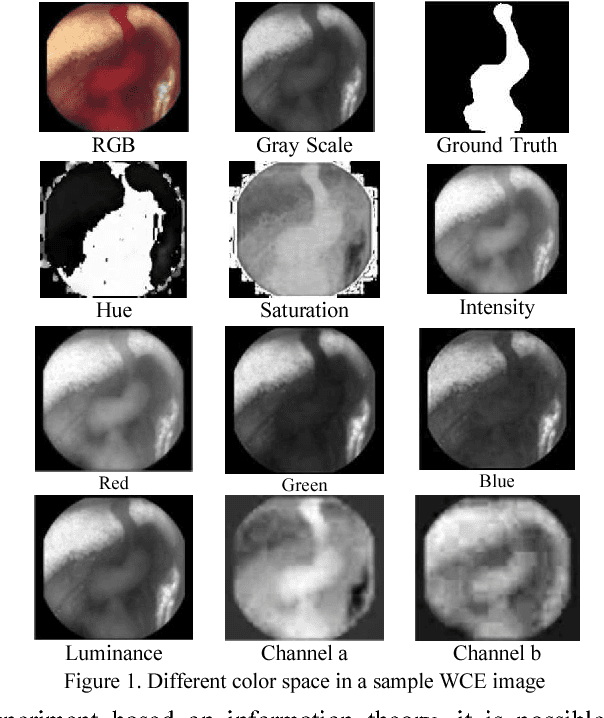
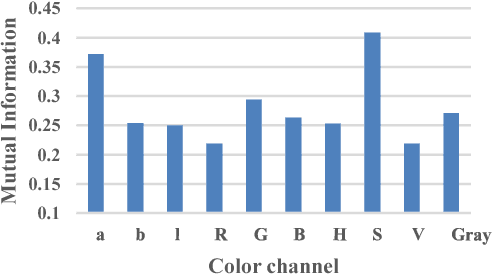
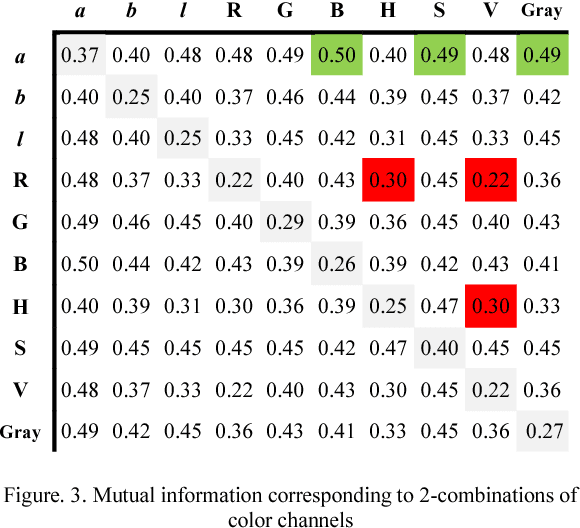
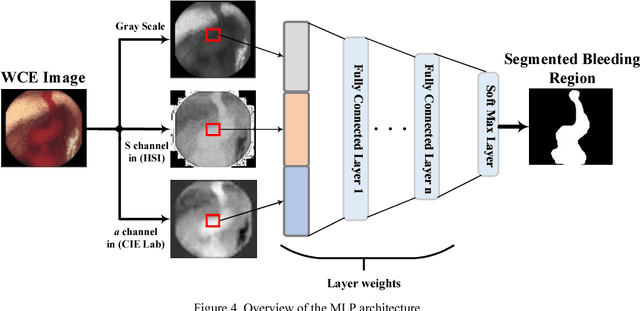
Abstract:Wireless capsule endoscopy (WCE) is an effective mean for diagnosis of gastrointestinal disorders. Detection of informative scenes in WCE video could reduce the length of transmitted videos and help the diagnosis procedure. In this paper, we investigate the problem of simplification of neural networks for automatic bleeding region detection inside capsule endoscopy device. Suitable color channels are selected as neural networks inputs, and image classification is conducted using a multi-layer perceptron (MLP) and a convolutional neural network (CNN) separately. Both CNN and MLP structures are simplified to reduce the number of computational operations. Performances of two simplified networks are evaluated on a WCE bleeding image dataset using the DICE score. Simulation results show that applying simplification methods on both MLP and CNN structures reduces the number of computational operations significantly with AUC greater than 0.97. Although CNN performs better in comparison with simplified MLP, the simplified MLP segments bleeding regions with a significantly smaller number of computational operations. Concerning the importance of having a simple structure or a more accurate model, each of the designed structures could be selected for inside capsule implementation.
Dense Fully Convolutional Network for Skin Lesion Segmentation
Jun 05, 2018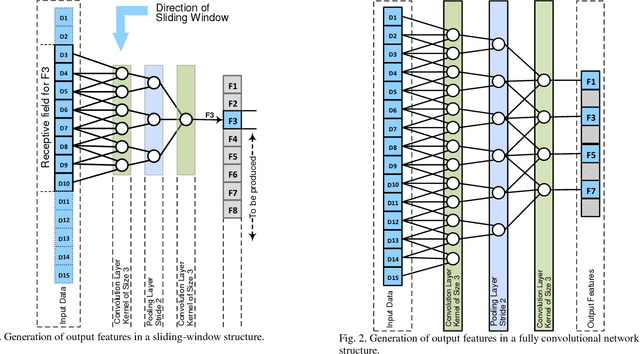

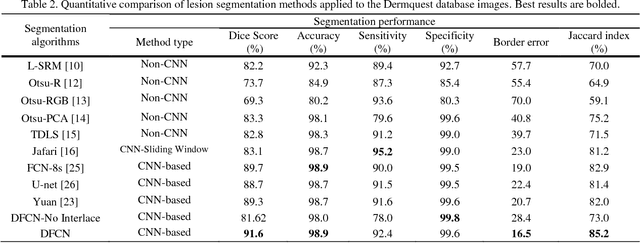
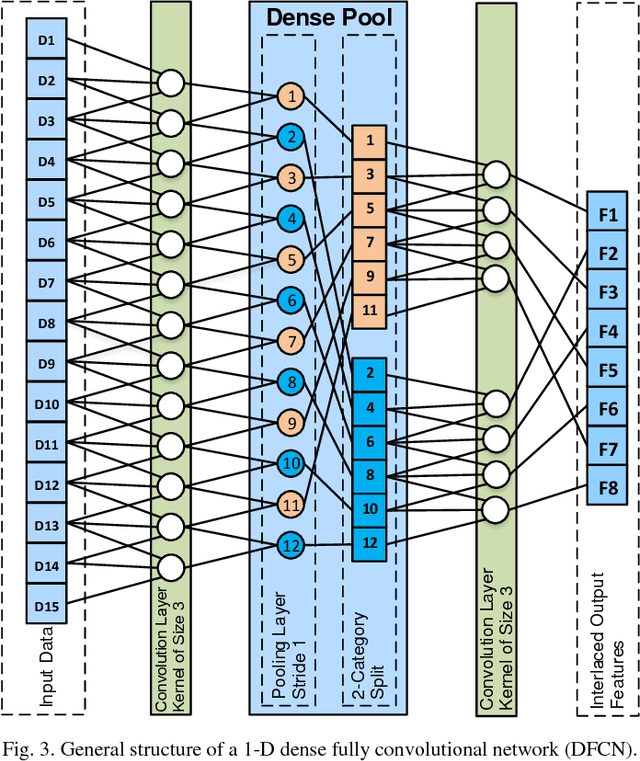
Abstract:Lesion segmentation in skin images is an important step in computerized detection of skin cancer. Melanoma is known as one of the most life threatening types of this cancer. Existing methods often fall short of accurately segmenting lesions with fuzzy boarders. In this paper, a new class of fully convolutional network is proposed, with new dense pooling layers for segmentation of lesion regions in non-dermoscopic images. Unlike other existing convolutional networks, this proposed network is designed to produce dense feature maps. This network leads to highly accurate segmentation of lesions. The produced dice score here is 91.6% which outperforms state-of-the-art algorithms in segmentation of skin lesions based on the Dermquest dataset.
Deep Learning in Pharmacogenomics: From Gene Regulation to Patient Stratification
Mar 07, 2018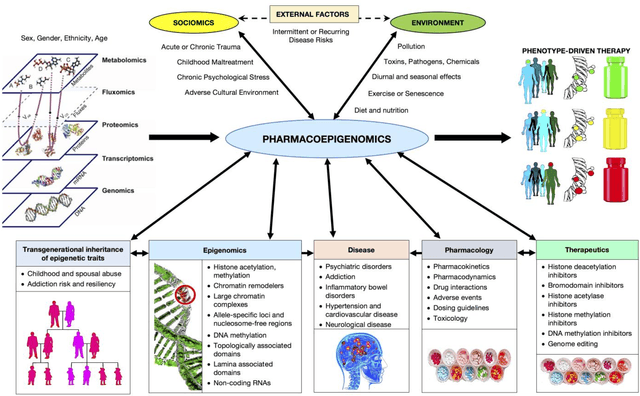
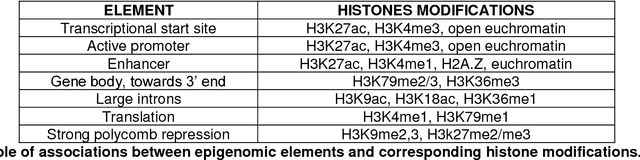
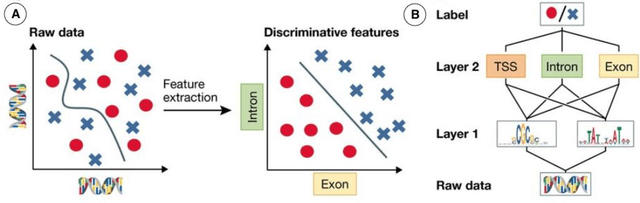
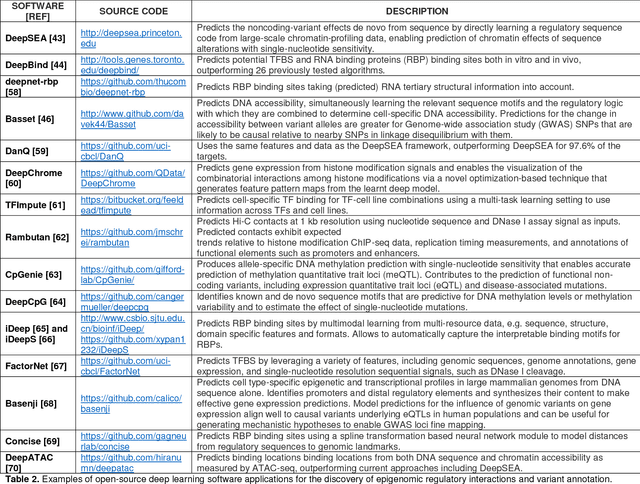
Abstract:This Perspective provides examples of current and future applications of deep learning in pharmacogenomics, including: (1) identification of novel regulatory variants located in noncoding domains and their function as applied to pharmacoepigenomics; (2) patient stratification from medical records; and (3) prediction of drugs, targets, and their interactions. Deep learning encapsulates a family of machine learning algorithms that over the last decade has transformed many important subfields of artificial intelligence (AI) and has demonstrated breakthrough performance improvements on a wide range of tasks in biomedicine. We anticipate that in the future deep learning will be widely used to predict personalized drug response and optimize medication selection and dosing, using knowledge extracted from large and complex molecular, epidemiological, clinical, and demographic datasets.
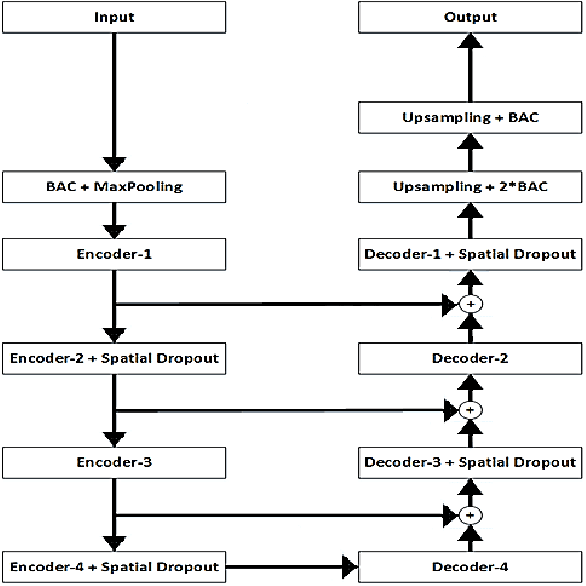
Liver segmentation in CT images using three dimensional to two dimensional fully convolutional network
Mar 03, 2018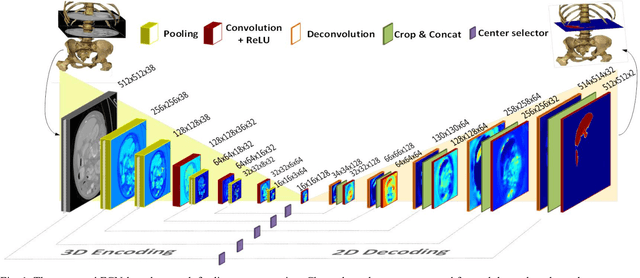
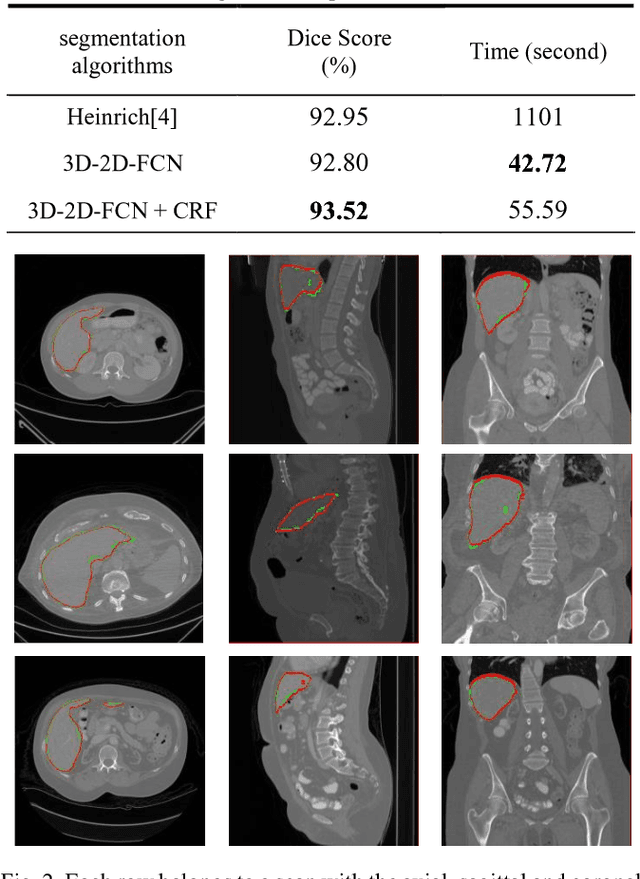
Abstract:The need for CT scan analysis is growing for pre-diagnosis and therapy of abdominal organs. Automatic organ segmentation of abdominal CT scan can help radiologists analyze the scans faster and segment organ images with fewer errors. However, existing methods are not efficient enough to perform the segmentation process for victims of accidents and emergencies situations. In this paper we propose an efficient liver segmentation with our 3D to 2D fully connected network (3D-2D-FCN). The segmented mask is enhanced by means of conditional random field on the organ's border. Consequently, we segment a target liver in less than a minute with Dice score of 93.52.
Adaptive specular reflection detection and inpainting in colonoscopy video frames
Feb 23, 2018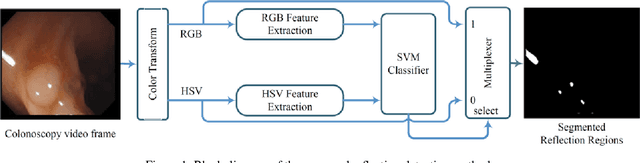

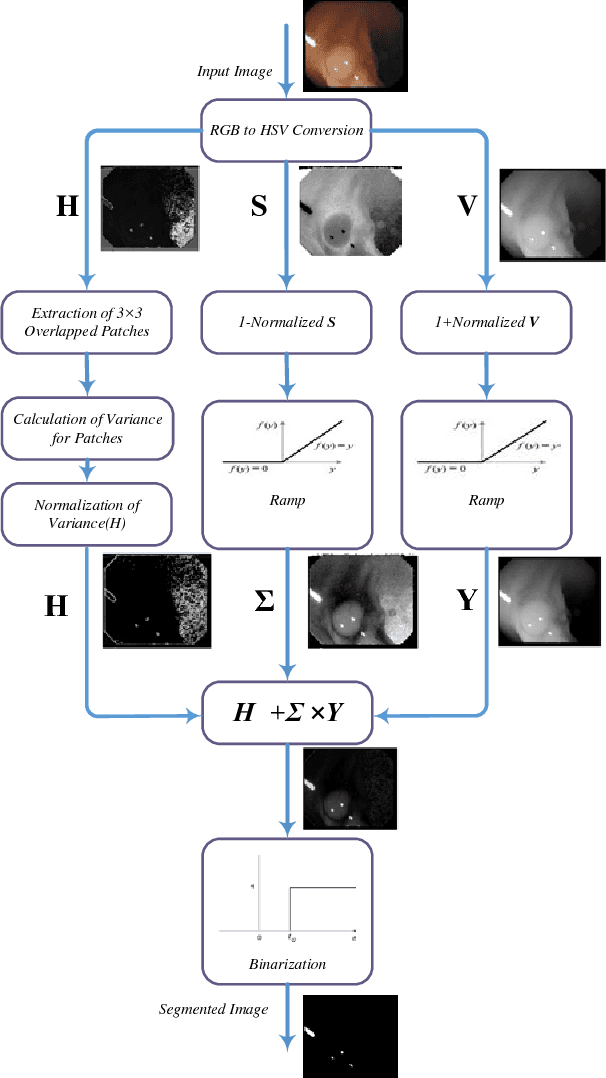
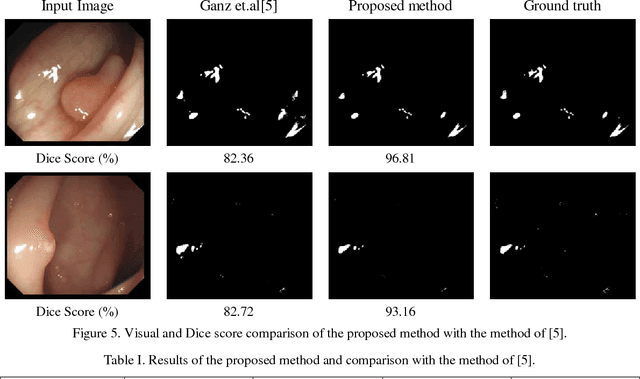
Abstract:Colonoscopy video frames might be contaminated by bright spots with unsaturated values known as specular reflection. Detection and removal of such reflections could enhance the quality of colonoscopy images and facilitate diagnosis procedure. In this paper we propose a novel two-phase method for this purpose, consisting of detection and removal phases. In the detection phase, we employ both HSV and RGB color space information for segmentation of specular reflections. We first train a non-linear SVM for selecting a color space based on image statistical features extracted from each channel of the color spaces. Then, a cost function for detection of specular reflections is introduced. In the removal phase, we propose a two-step inpainting method which consists of appropriate replacement patch selection and removal of the blockiness effects. The proposed method is evaluated by testing on an available colonoscopy image database where accuracy and Dice score of 99.68% and 71.79% are achieved respectively.
Left ventricle segmentation By modelling uncertainty in prediction of deep convolutional neural networks and adaptive thresholding inference
Feb 23, 2018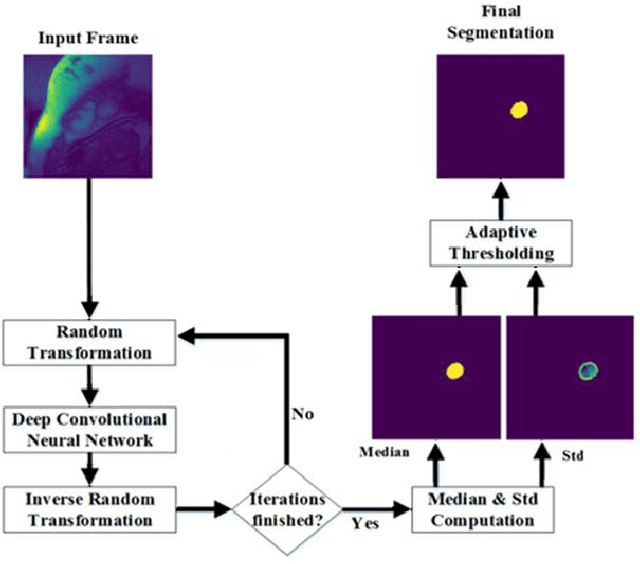
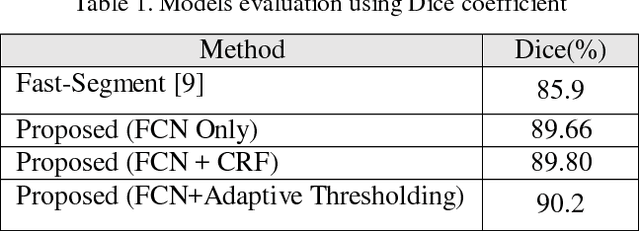

Abstract:Deep neural networks have shown great achievements in solving complex problems. However, there are fundamental problems that limit their real world applications. Lack of measurable criteria for estimating uncertainty in the network outputs is one of these problems. In this paper, we address this limitation by introducing deformation to the network input and measuring the level of stability in the network's output. We calculate simple random transformations to estimate the prediction uncertainty of deep convolutional neural networks. For a real use-case, we apply this method to left ventricle segmentation in MRI cardiac images. We also propose an adaptive thresholding method to consider the deep neural network uncertainty. Experimental results demonstrate state-of-the-art performance and highlight the capabilities of simple methods in conjunction with deep neural networks.
Liver Segmentation in Abdominal CT Images by Adaptive 3D Region Growing
Feb 21, 2018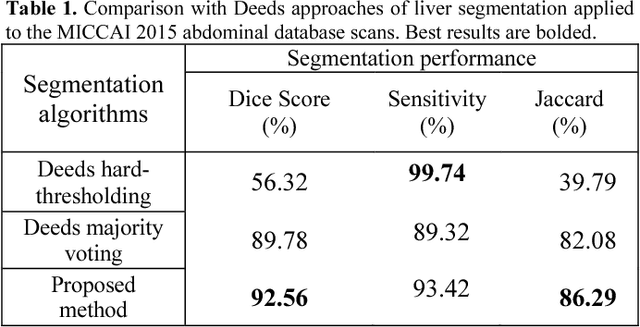
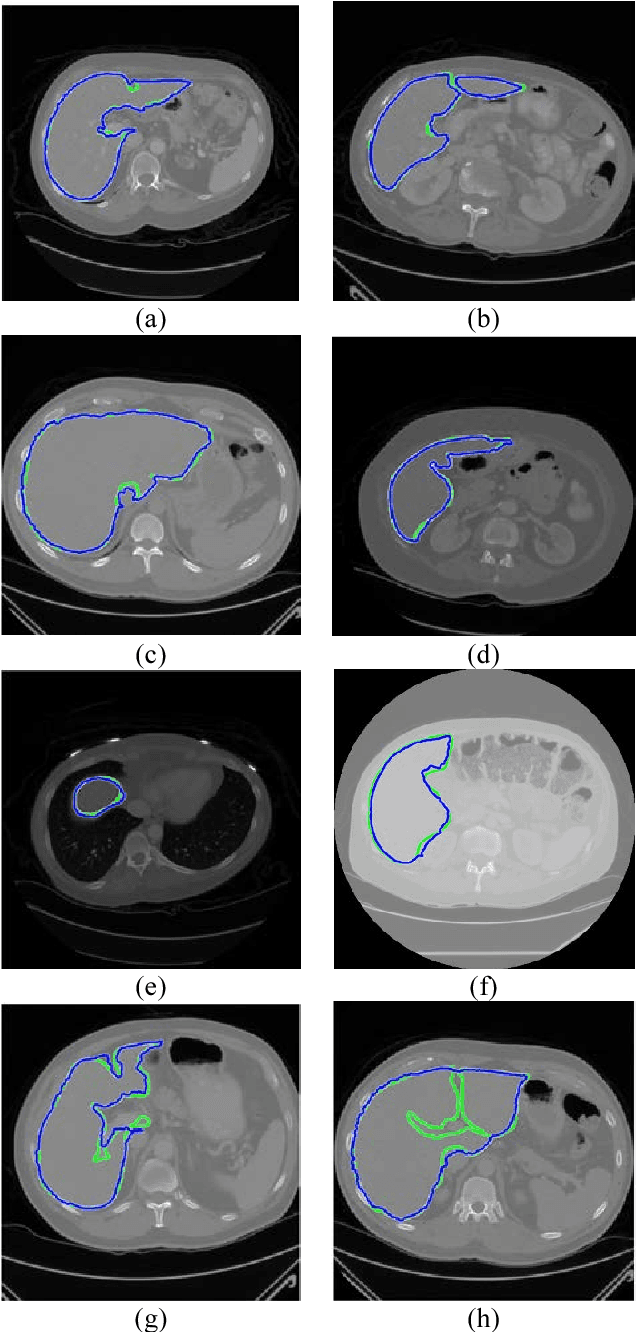
Abstract:Automatic liver segmentation plays an important role in computer-aided diagnosis and treatment. Manual segmentation of organs is a difficult and tedious task and so prone to human errors. In this paper, we propose an adaptive 3D region growing with subject-specific conditions. For this aim we use the intensity distribution of most probable voxels in prior map along with location prior. We also incorporate the boundary of target organs to restrict the region growing. In order to obtain strong edges and high contrast, we propose an effective contrast enhancement algorithm to facilitate more accurate segmentation. In this paper, 92.56% Dice score is achieved. We compare our method with the method of hard thresholding on Deeds prior map and also with the majority voting on Deeds registration with 13 organs.
Segmentation of Bleeding Regions in Wireless Capsule Endoscopy Images an Approach for inside Capsule Video Summarization
Feb 21, 2018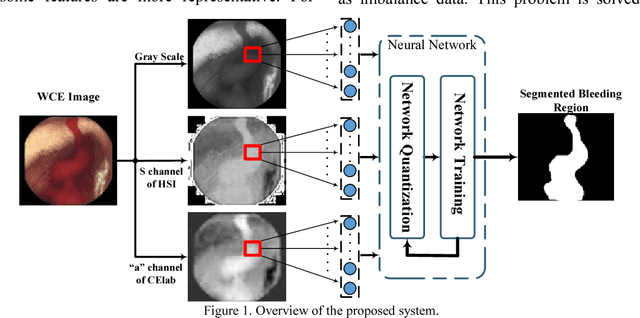
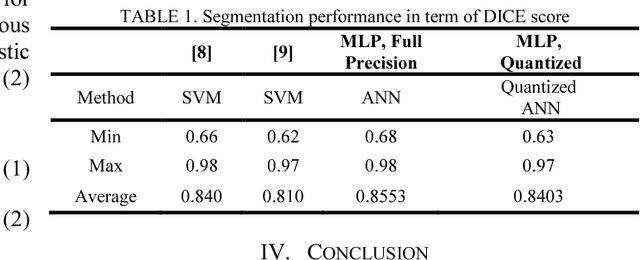
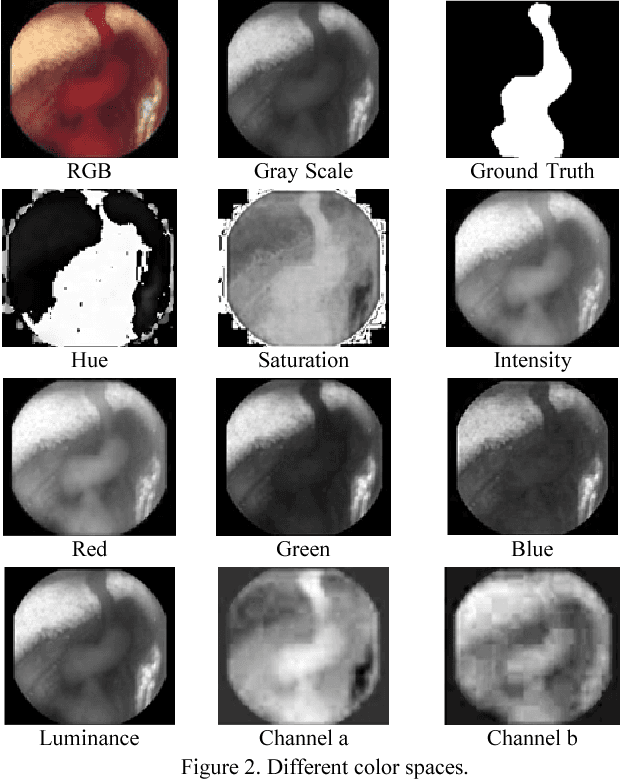
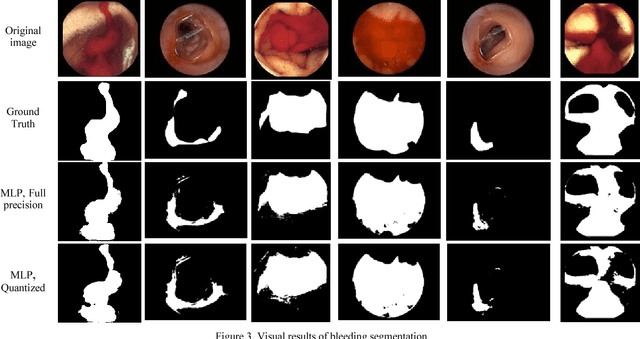
Abstract:Wireless capsule endoscopy (WCE) is an effective means of diagnosis of gastrointestinal disorders. Detection of informative scenes by WCE could reduce the length of transmitted videos and can help with the diagnosis. In this paper we propose a simple and efficient method for segmentation of the bleeding regions in WCE captured images. Suitable color channels are selected and classified by a multi-layer perceptron (MLP) structure. The MLP structure is quantized such that the implementation does not require multiplications. The proposed method is tested by simulation on WCE bleeding image dataset. The proposed structure is designed considering hardware resource constrains that exist in WCE systems.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge