Flavio D. Colavecchia
Automatic quantification of the LV function and mass: a deep learning approach for cardiovascular MRI
Dec 14, 2018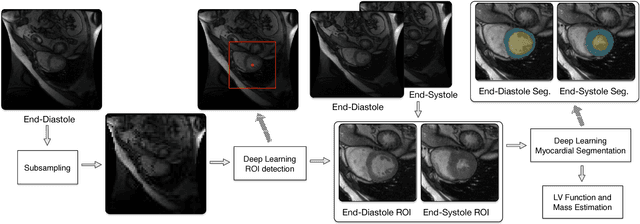
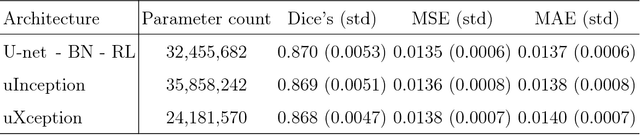
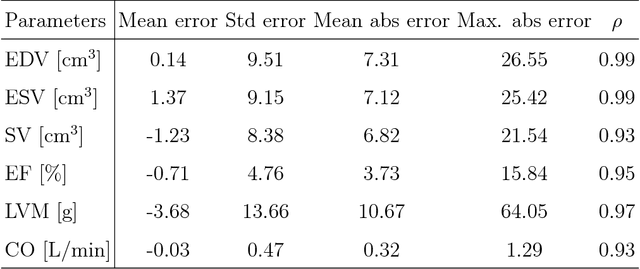
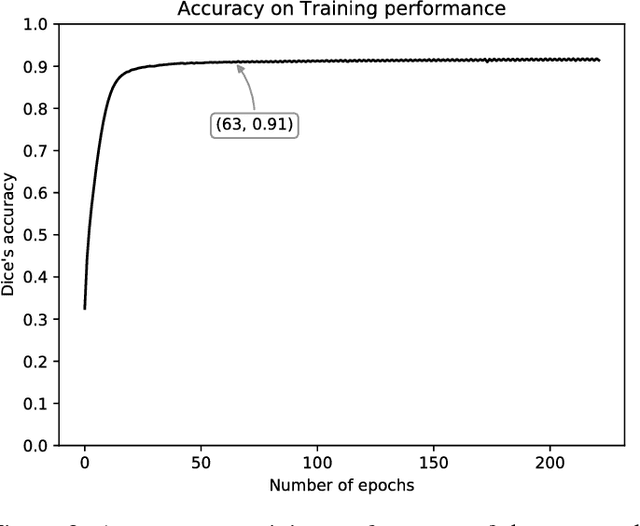
Abstract:Objective: This paper proposes a novel approach for automatic left ventricle (LV) quantification using convolutional neural networks (CNN). Methods: The general framework consists of one CNN for detecting the LV, and another for tissue classification. Also, three new deep learning architectures were proposed for LV quantification. These new CNNs introduce the ideas of sparsity and depthwise separable convolution into the U-net architecture, as well as, a residual learning strategy level-to-level. To this end, we extend the classical U-net architecture and use the generalized Jaccard distance as optimization objective function. Results: The CNNs were trained and evaluated with 140 patients from two public cardiovascular magnetic resonance datasets (Sunnybrook and Cardiac Atlas Project) by using a 5-fold cross-validation strategy. Our results demonstrate a suitable accuracy for myocardial segmentation ($\sim$0.9 Dice's coefficient), and a strong correlation with the most relevant physiological measures: 0.99 for end-diastolic and end-systolic volume, 0.97 for the left myocardial mass, 0.95 for the ejection fraction and 0.93 for the stroke volume and cardiac output. Conclusion: Our simulation and clinical evaluation results demonstrate the capability and merits of the proposed CNN to estimate different structural and functional features such as LV mass and EF which are commonly used for both diagnosis and treatment of different pathologies. Significance: This paper suggests a new approach for automatic LV quantification based on deep learning where errors are comparable to the inter- and intra-operator ranges for manual contouring. Also, this approach may have important applications on motion quantification.
Automatic Myocardial Segmentation by Using A Deep Learning Network in Cardiac MRI
Aug 24, 2017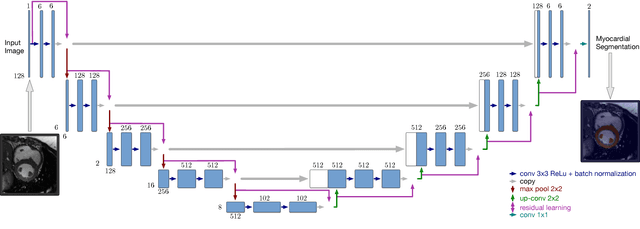
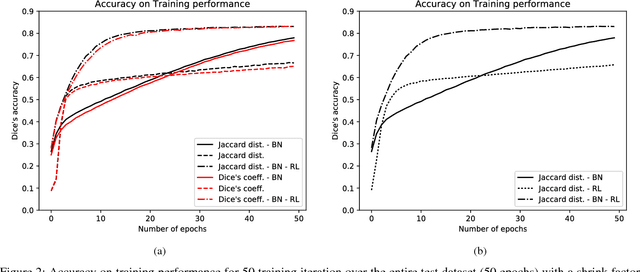

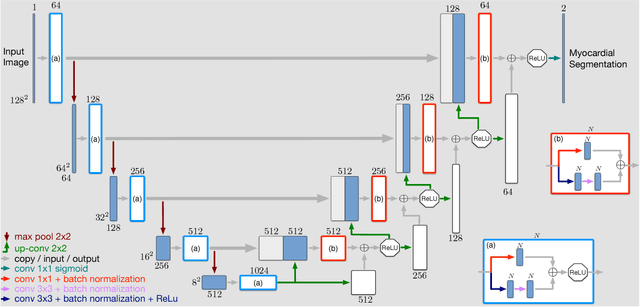
Abstract:Cardiac function is of paramount importance for both prognosis and treatment of different pathologies such as mitral regurgitation, ischemia, dyssynchrony and myocarditis. Cardiac behavior is determined by structural and functional features. In both cases, the analysis of medical imaging studies requires to detect and segment the myocardium. Nowadays, magnetic resonance imaging (MRI) is one of the most relevant and accurate non-invasive diagnostic tools for cardiac structure and function. In this work we propose to use a deep learning technique to assist the automatization of myocardial segmentation in cardiac MRI. We present several improvements to previous works in this paper: we propose to use the Jaccard distance as optimization objective function, we integrate a residual learning strategy into the code, and we introduce a batch normalization layer to train the fully convolutional neural network. Our results demonstrate that this architecture outperforms previous approaches based on a similar network architecture, and that provides a suitable approach for myocardial segmentation. Our benchmark shows that the automatic myocardial segmentation takes less than 22 seg. for a volume of 128~x~128~x~13 pixels in a 3.1 GHz intel core i7.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge