Zeynel A. Samak
Automatic Prediction of Stroke Treatment Outcomes: Latest Advances and Perspectives
Dec 06, 2024Abstract:Stroke is a major global health problem that causes mortality and morbidity. Predicting the outcomes of stroke intervention can facilitate clinical decision-making and improve patient care. Engaging and developing deep learning techniques can help to analyse large and diverse medical data, including brain scans, medical reports and other sensor information, such as EEG, ECG, EMG and so on. Despite the common data standardisation challenge within medical image analysis domain, the future of deep learning in stroke outcome prediction lie in using multimodal information, including final infarct data, to achieve better prediction of long-term functional outcomes. This article provides a broad review of recent advances and applications of deep learning in the prediction of stroke outcomes, including (i) the data and models used, (ii) the prediction tasks and measures of success, (iii) the current challenges and limitations, and (iv) future directions and potential benefits. This comprehensive review aims to provide researchers, clinicians, and policy makers with an up-to-date understanding of this rapidly evolving and promising field.
TranSOP: Transformer-based Multimodal Classification for Stroke Treatment Outcome Prediction
Jan 25, 2023Abstract:Acute ischaemic stroke, caused by an interruption in blood flow to brain tissue, is a leading cause of disability and mortality worldwide. The selection of patients for the most optimal ischaemic stroke treatment is a crucial step for a successful outcome, as the effect of treatment highly depends on the time to treatment. We propose a transformer-based multimodal network (TranSOP) for a classification approach that employs clinical metadata and imaging information, acquired on hospital admission, to predict the functional outcome of stroke treatment based on the modified Rankin Scale (mRS). This includes a fusion module to efficiently combine 3D non-contrast computed tomography (NCCT) features and clinical information. In comparative experiments using unimodal and multimodal data on the MRCLEAN dataset, we achieve a state-of-the-art AUC score of 0.85.
Prediction of Thrombectomy Functional Outcomes using Multimodal Data
May 28, 2020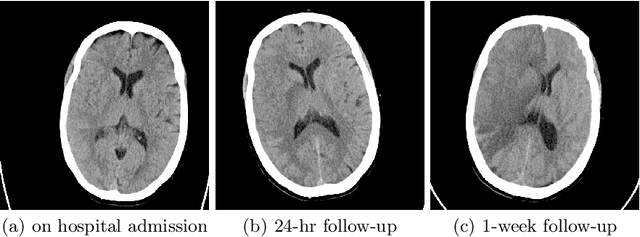
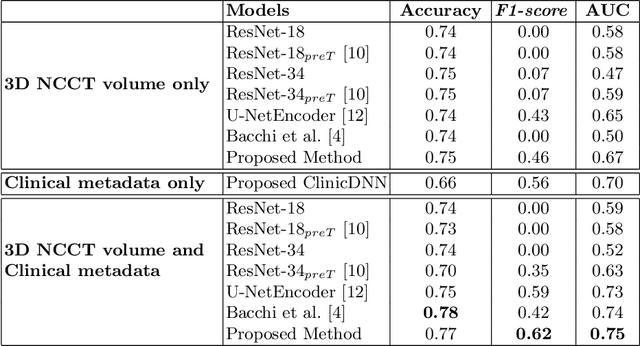
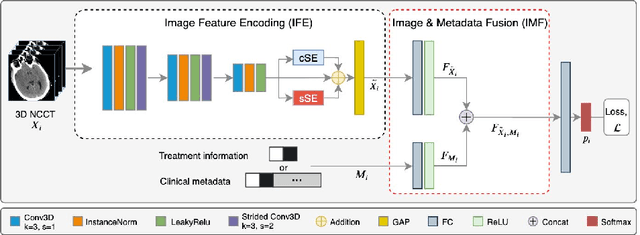
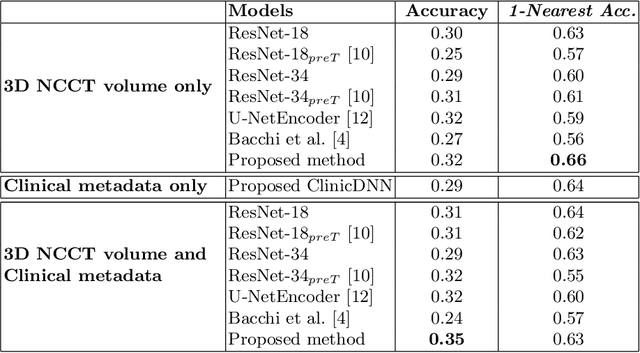
Abstract:Recent randomised clinical trials have shown that patients with ischaemic stroke {due to occlusion of a large intracranial blood vessel} benefit from endovascular thrombectomy. However, predicting outcome of treatment in an individual patient remains a challenge. We propose a novel deep learning approach to directly exploit multimodal data (clinical metadata information, imaging data, and imaging biomarkers extracted from images) to estimate the success of endovascular treatment. We incorporate an attention mechanism in our architecture to model global feature inter-dependencies, both channel-wise and spatially. We perform comparative experiments using unimodal and multimodal data, to predict functional outcome (modified Rankin Scale score, mRS) and achieve 0.75 AUC for dichotomised mRS scores and 0.35 classification accuracy for individual mRS scores.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge