Masoom A. Haider
ProstateGAN: Mitigating Data Bias via Prostate Diffusion Imaging Synthesis with Generative Adversarial Networks
Nov 21, 2018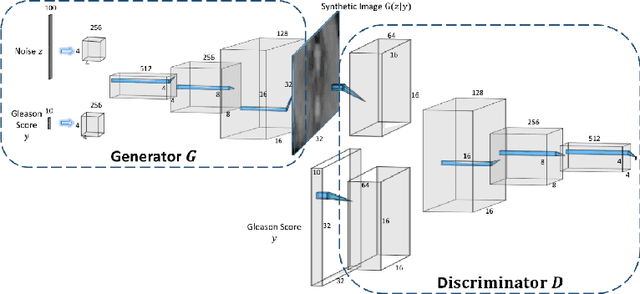

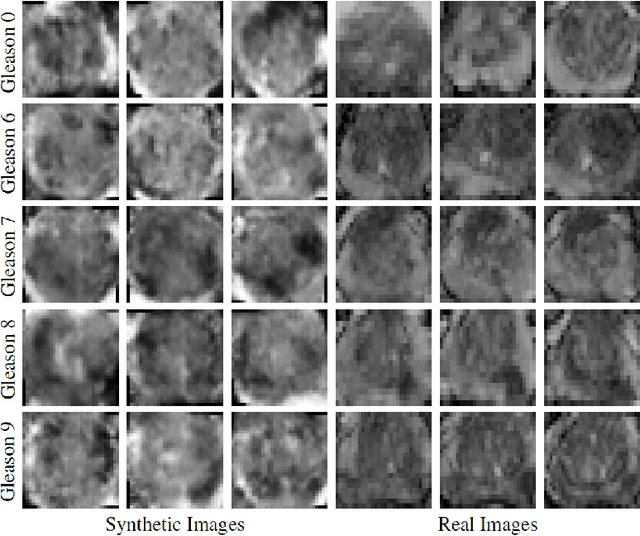
Abstract:Generative Adversarial Networks (GANs) have shown considerable promise for mitigating the challenge of data scarcity when building machine learning-driven analysis algorithms. Specifically, a number of studies have shown that GAN-based image synthesis for data augmentation can aid in improving classification accuracy in a number of medical image analysis tasks, such as brain and liver image analysis. However, the efficacy of leveraging GANs for tackling prostate cancer analysis has not been previously explored. Motivated by this, in this study we introduce ProstateGAN, a GAN-based model for synthesizing realistic prostate diffusion imaging data. More specifically, in order to generate new diffusion imaging data corresponding to a particular cancer grade (Gleason score), we propose a conditional deep convolutional GAN architecture that takes Gleason scores into consideration during the training process. Experimental results show that high-quality synthetic prostate diffusion imaging data can be generated using the proposed ProstateGAN for specified Gleason scores.
Discovery Radiomics via Evolutionary Deep Radiomic Sequencer Discovery for Pathologically-Proven Lung Cancer Detection
Oct 20, 2017Abstract:While lung cancer is the second most diagnosed form of cancer in men and women, a sufficiently early diagnosis can be pivotal in patient survival rates. Imaging-based, or radiomics-driven, detection methods have been developed to aid diagnosticians, but largely rely on hand-crafted features which may not fully encapsulate the differences between cancerous and healthy tissue. Recently, the concept of discovery radiomics was introduced, where custom abstract features are discovered from readily available imaging data. We propose a novel evolutionary deep radiomic sequencer discovery approach based on evolutionary deep intelligence. Motivated by patient privacy concerns and the idea of operational artificial intelligence, the evolutionary deep radiomic sequencer discovery approach organically evolves increasingly more efficient deep radiomic sequencers that produce significantly more compact yet similarly descriptive radiomic sequences over multiple generations. As a result, this framework improves operational efficiency and enables diagnosis to be run locally at the radiologist's computer while maintaining detection accuracy. We evaluated the evolved deep radiomic sequencer (EDRS) discovered via the proposed evolutionary deep radiomic sequencer discovery framework against state-of-the-art radiomics-driven and discovery radiomics methods using clinical lung CT data with pathologically-proven diagnostic data from the LIDC-IDRI dataset. The evolved deep radiomic sequencer shows improved sensitivity (93.42%), specificity (82.39%), and diagnostic accuracy (88.78%) relative to previous radiomics approaches.
Discovery Radiomics for Pathologically-Proven Computed Tomography Lung Cancer Prediction
Mar 28, 2017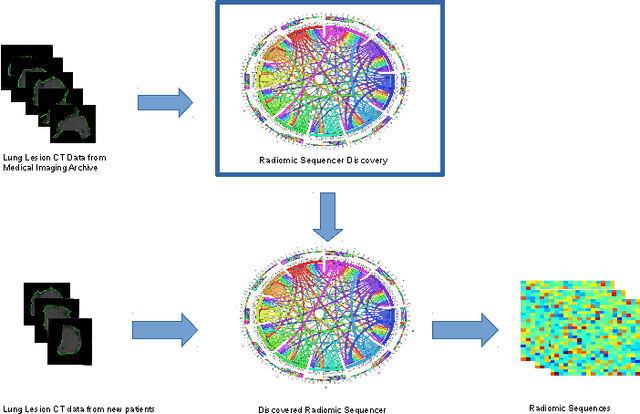
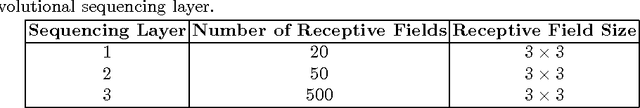
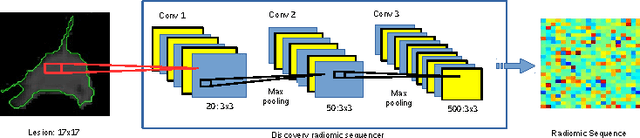
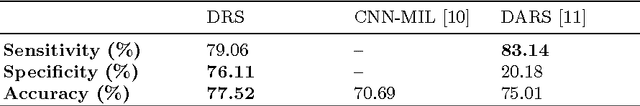
Abstract:Lung cancer is the leading cause for cancer related deaths. As such, there is an urgent need for a streamlined process that can allow radiologists to provide diagnosis with greater efficiency and accuracy. A powerful tool to do this is radiomics: a high-dimension imaging feature set. In this study, we take the idea of radiomics one step further by introducing the concept of discovery radiomics for lung cancer prediction using CT imaging data. In this study, we realize these custom radiomic sequencers as deep convolutional sequencers using a deep convolutional neural network learning architecture. To illustrate the prognostic power and effectiveness of the radiomic sequences produced by the discovered sequencer, we perform cancer prediction between malignant and benign lesions from 97 patients using the pathologically-proven diagnostic data from the LIDC-IDRI dataset. Using the clinically provided pathologically-proven data as ground truth, the proposed framework provided an average accuracy of 77.52% via 10-fold cross-validation with a sensitivity of 79.06% and specificity of 76.11%, surpassing the state-of-the art method.
Noise-Compensated, Bias-Corrected Diffusion Weighted Endorectal Magnetic Resonance Imaging via a Stochastically Fully-Connected Joint Conditional Random Field Model
Jul 05, 2016
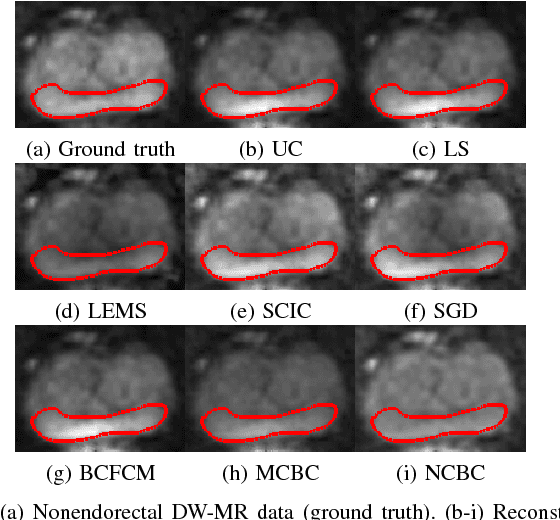
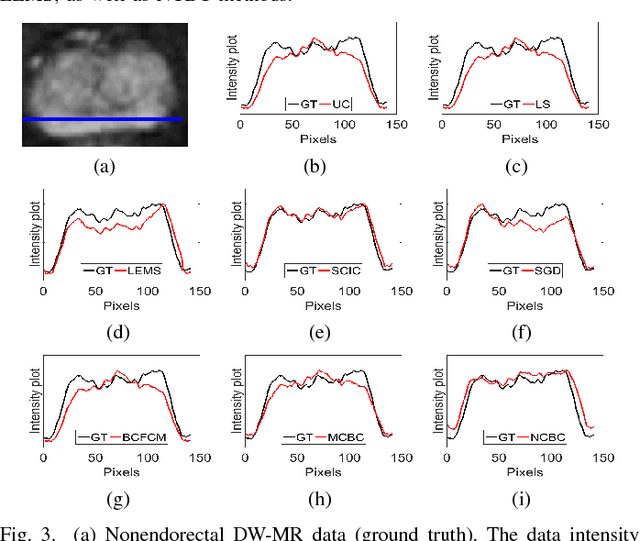
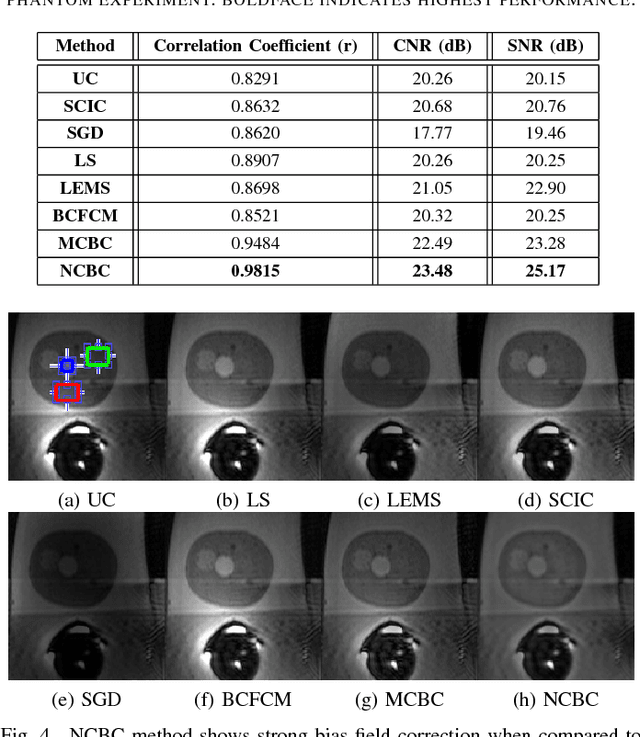
Abstract:Diffusion weighted magnetic resonance imaging (DW-MR) is a powerful tool in imaging-based prostate cancer screening and detection. Endorectal coils are commonly used in DW-MR imaging to improve the signal-to-noise ratio (SNR) of the acquisition, at the expense of significant intensity inhomogeneities (bias field) that worsens as we move away from the endorectal coil. The presence of bias field can have a significant negative impact on the accuracy of different image analysis tasks, as well as prostate tumor localization, thus leading to increased inter- and intra-observer variability. Retrospective bias correction approaches are introduced as a more efficient way of bias correction compared to the prospective methods such that they correct for both of the scanner and anatomy-related bias fields in MR imaging. Previously proposed retrospective bias field correction methods suffer from undesired noise amplification that can reduce the quality of bias-corrected DW-MR image. Here, we propose a unified data reconstruction approach that enables joint compensation of bias field as well as data noise in DW-MR imaging. The proposed noise-compensated, bias-corrected (NCBC) data reconstruction method takes advantage of a novel stochastically fully connected joint conditional random field (SFC-JCRF) model to mitigate the effects of data noise and bias field in the reconstructed MR data. The proposed NCBC reconstruction method was tested on synthetic DW-MR data, physical DW-phantom as well as real DW-MR data all acquired using endorectal MR coil. Both qualitative and quantitative analysis illustrated that the proposed NCBC method can achieve improved image quality when compared to other tested bias correction methods. As such, the proposed NCBC method may have potential as a useful retrospective approach for improving the consistency of image interpretations.
Sparse Reconstruction of Compressive Sensing MRI using Cross-Domain Stochastically Fully Connected Conditional Random Fields
Dec 25, 2015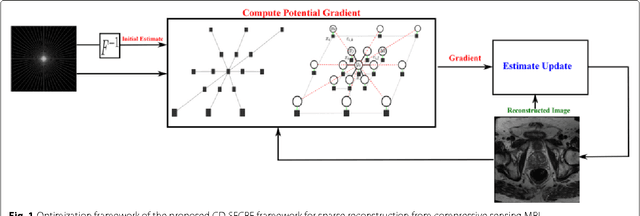


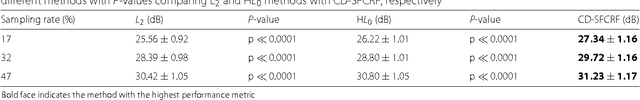
Abstract:Magnetic Resonance Imaging (MRI) is a crucial medical imaging technology for the screening and diagnosis of frequently occurring cancers. However image quality may suffer by long acquisition times for MRIs due to patient motion, as well as result in great patient discomfort. Reducing MRI acquisition time can reduce patient discomfort and as a result reduces motion artifacts from the acquisition process. Compressive sensing strategies, when applied to MRI, have been demonstrated to be effective at decreasing acquisition times significantly by sparsely sampling the \emph{k}-space during the acquisition process. However, such a strategy requires advanced reconstruction algorithms to produce high quality and reliable images from compressive sensing MRI. This paper proposes a new reconstruction approach based on cross-domain stochastically fully connected conditional random fields (CD-SFCRF) for compressive sensing MRI. The CD-SFCRF introduces constraints in both \emph{k}-space and spatial domains within a stochastically fully connected graphical model to produce improved MRI reconstruction. Experimental results using T2-weighted (T2w) imaging and diffusion-weighted imaging (DWI) of the prostate show strong performance in preserving fine details and tissue structures in the reconstructed images when compared to other tested methods even at low sampling rates.
Discovery Radiomics for Multi-Parametric MRI Prostate Cancer Detection
Oct 20, 2015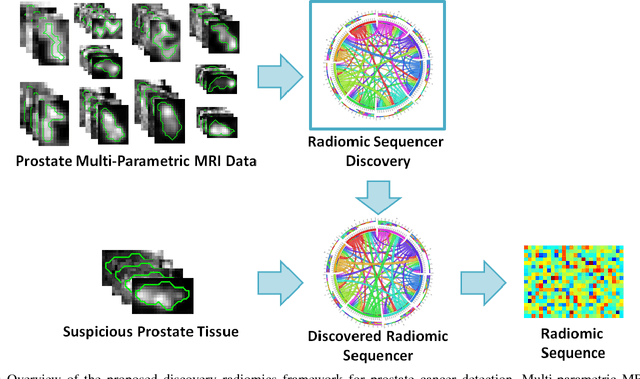
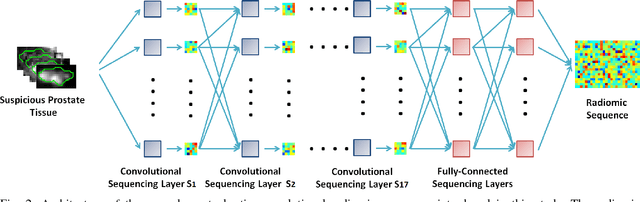
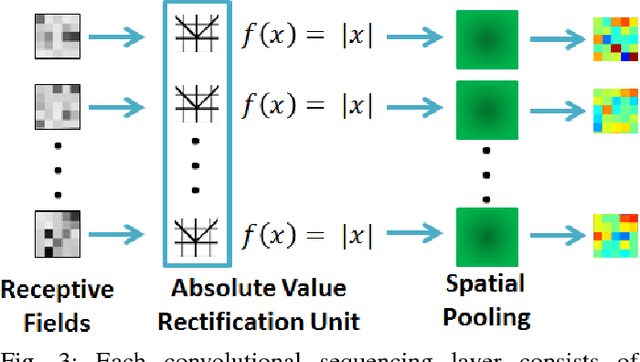

Abstract:Prostate cancer is the most diagnosed form of cancer in Canadian men, and is the third leading cause of cancer death. Despite these statistics, prognosis is relatively good with a sufficiently early diagnosis, making fast and reliable prostate cancer detection crucial. As imaging-based prostate cancer screening, such as magnetic resonance imaging (MRI), requires an experienced medical professional to extensively review the data and perform a diagnosis, radiomics-driven methods help streamline the process and has the potential to significantly improve diagnostic accuracy and efficiency, and thus improving patient survival rates. These radiomics-driven methods currently rely on hand-crafted sets of quantitative imaging-based features, which are selected manually and can limit their ability to fully characterize unique prostate cancer tumour phenotype. In this study, we propose a novel \textit{discovery radiomics} framework for generating custom radiomic sequences tailored for prostate cancer detection. Discovery radiomics aims to uncover abstract imaging-based features that capture highly unique tumour traits and characteristics beyond what can be captured using predefined feature models. In this paper, we discover new custom radiomic sequencers for generating new prostate radiomic sequences using multi-parametric MRI data. We evaluated the performance of the discovered radiomic sequencer against a state-of-the-art hand-crafted radiomic sequencer for computer-aided prostate cancer detection with a feedforward neural network using real clinical prostate multi-parametric MRI data. Results for the discovered radiomic sequencer demonstrate good performance in prostate cancer detection and clinical decision support relative to the hand-crafted radiomic sequencer. The use of discovery radiomics shows potential for more efficient and reliable automatic prostate cancer detection.
 Add to Chrome
Add to Chrome Add to Firefox
Add to Firefox Add to Edge
Add to Edge